Deposition Date
2025-05-15
Release Date
2025-11-19
Last Version Date
2025-12-03
Entry Detail
PDB ID:
9UYM
Keywords:
Title:
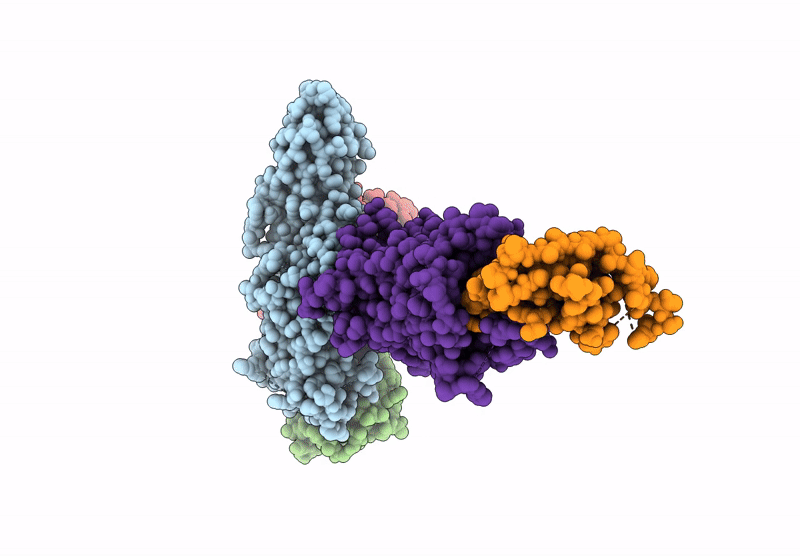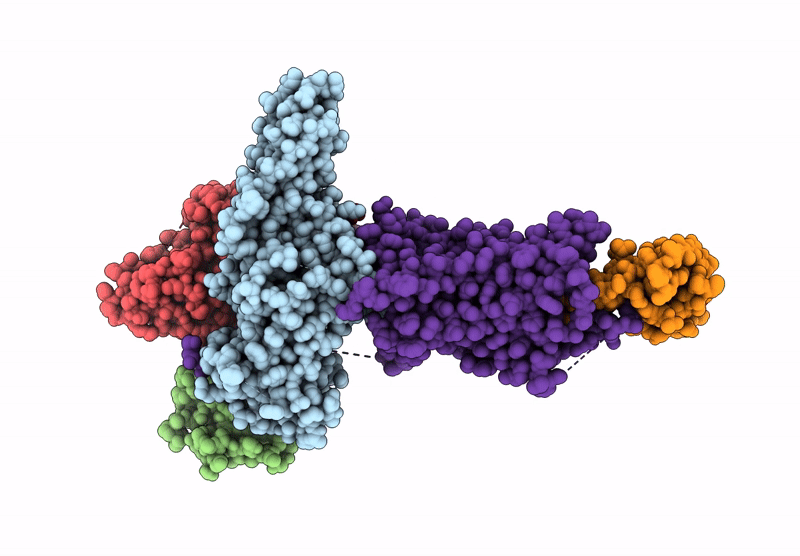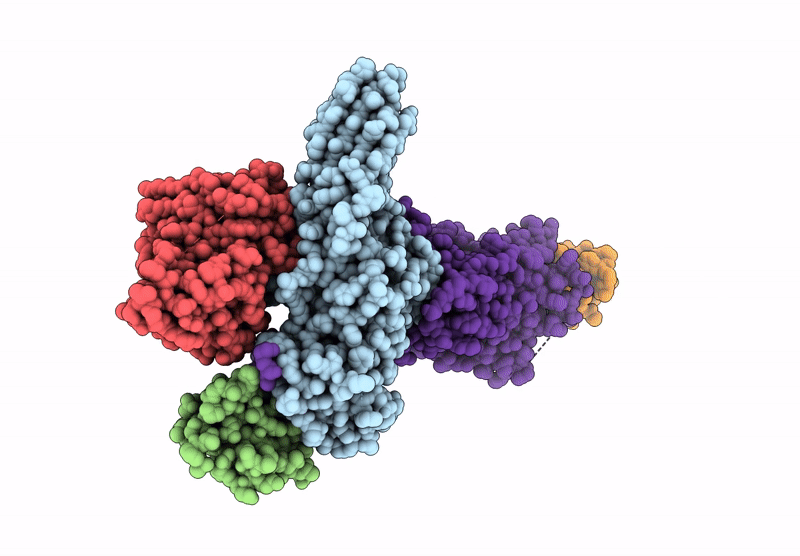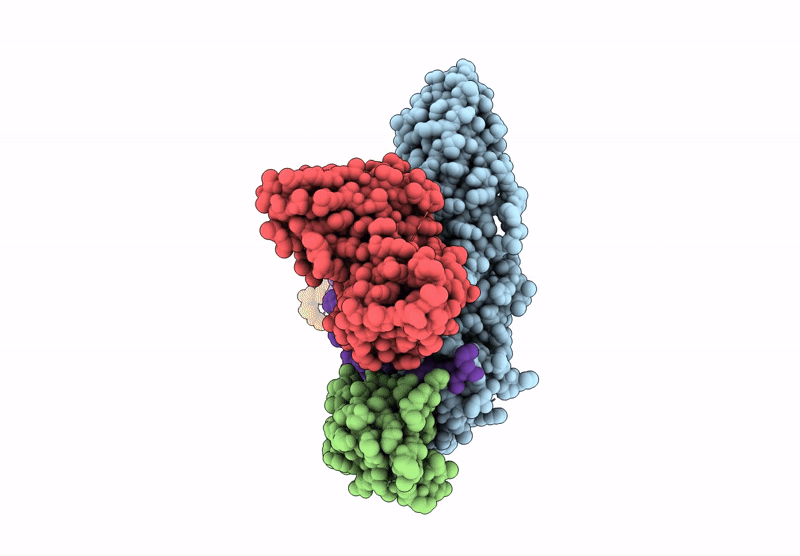
Composite map of the G protein-coupled receptor 1 (GPR1) bound to chemerin and beta-arrestin 2
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Escherichia phage EcSzw-2 (Taxon ID: 2419741)
Escherichia phage EcSzw-2 (Taxon ID: 2419741)
Expression System(s):
Method Details:
Experimental Method:
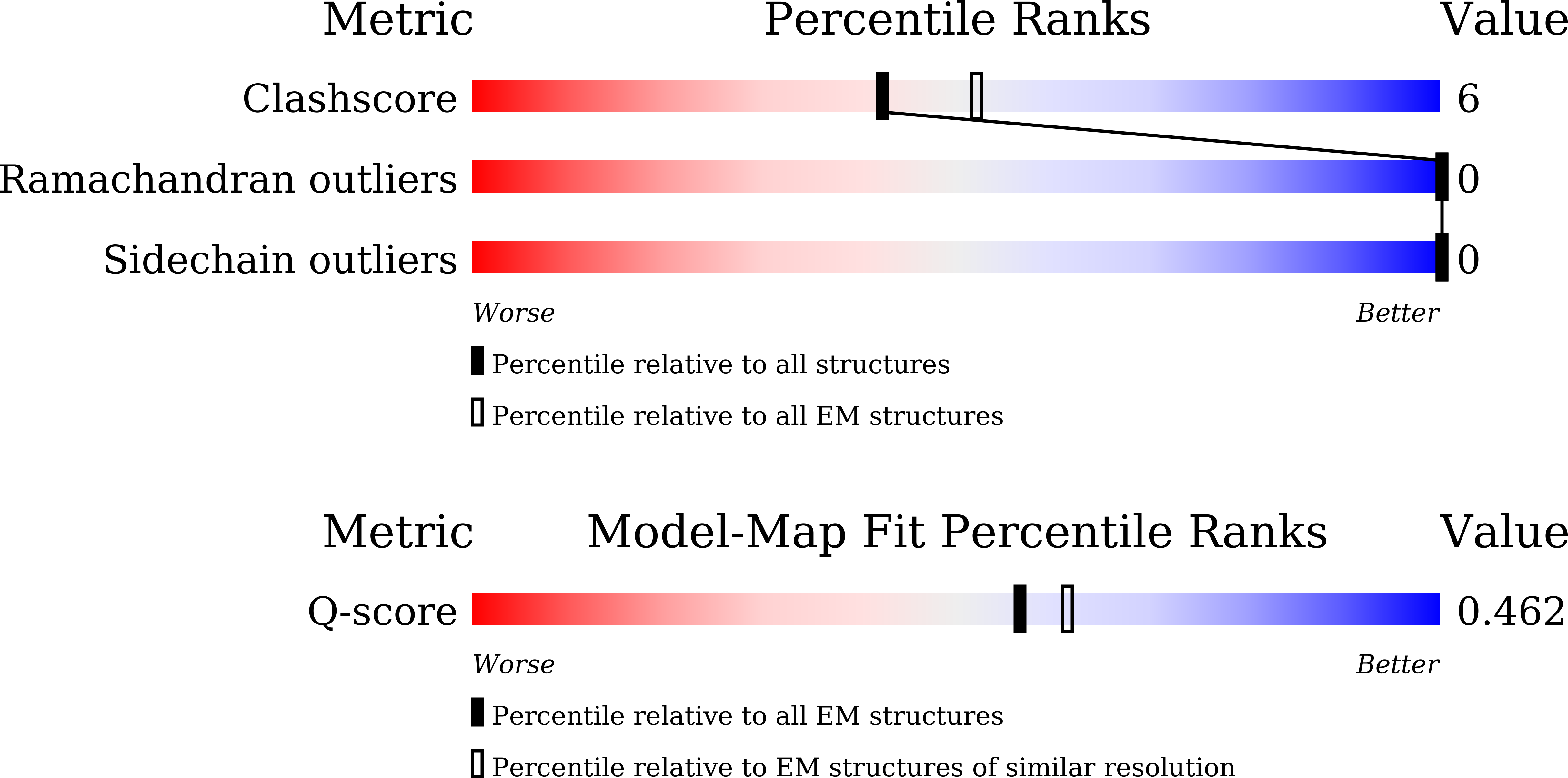
Resolution:
3.50 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE