Deposition Date
2025-03-26
Release Date
2026-02-11
Last Version Date
2026-02-11
Entry Detail
Biological Source:
Source Organism(s):
Enterococcus casseliflavus (Taxon ID: 37734)
Expression System(s):
Method Details:
Experimental Method:
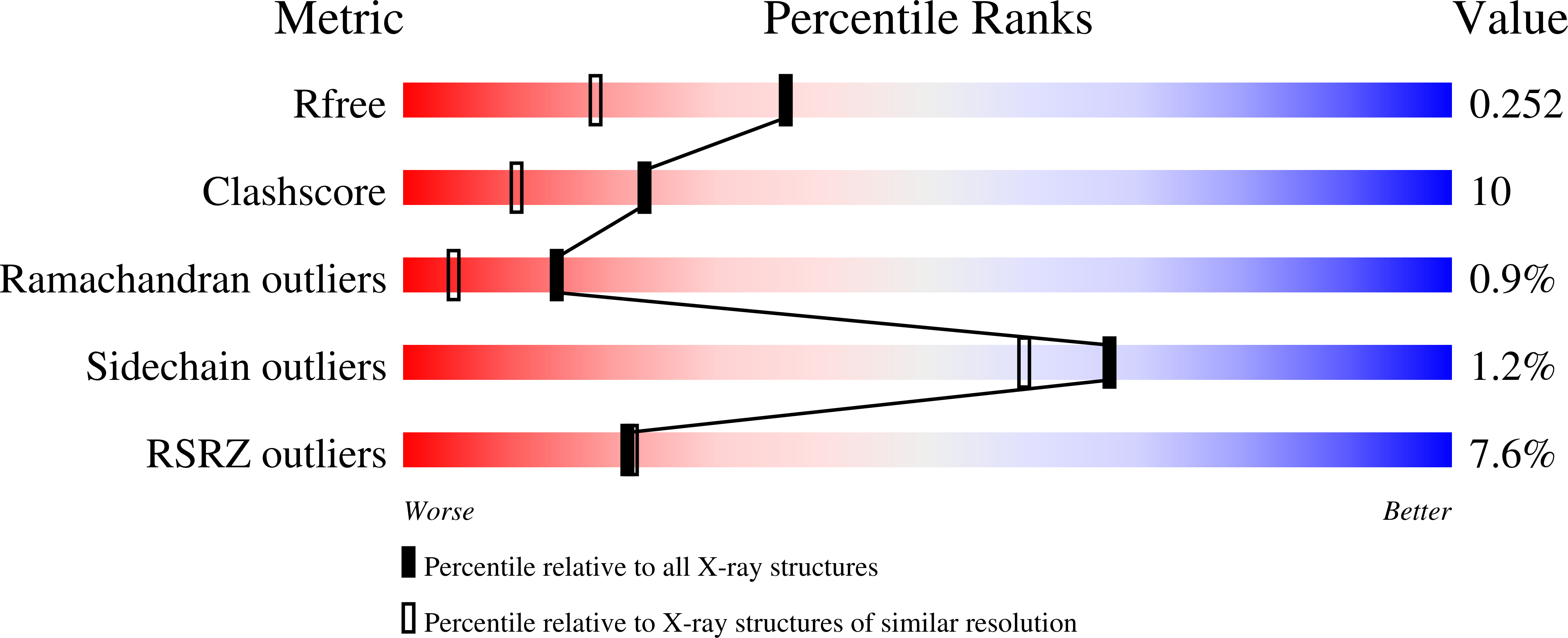
Resolution:
1.86 Å
R-Value Free:
0.25
R-Value Work:
0.21
R-Value Observed:
0.21
Space Group:
P 1 21 1