Deposition Date
2025-04-01
Release Date
2025-11-05
Last Version Date
2026-02-11
Entry Detail
PDB ID:
9NZM
Keywords:
Title:
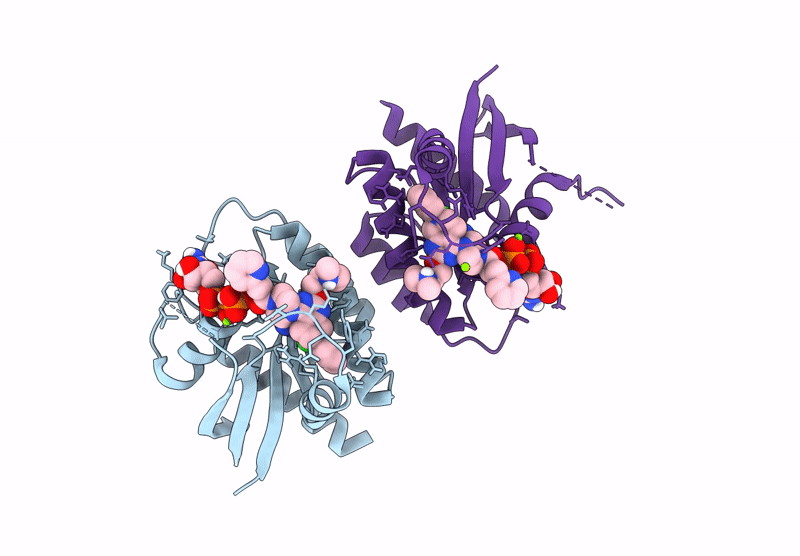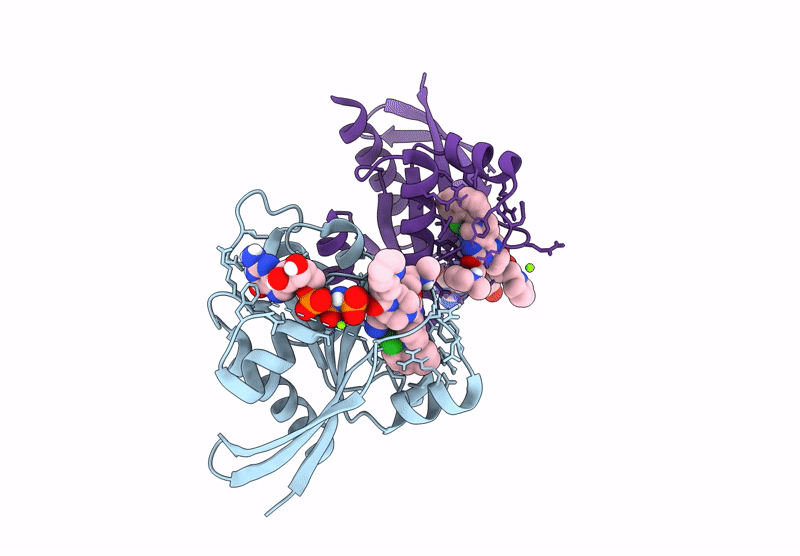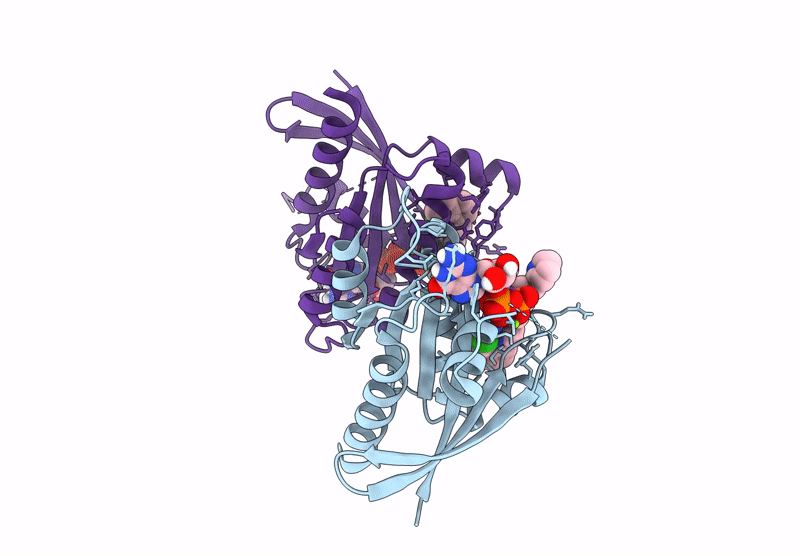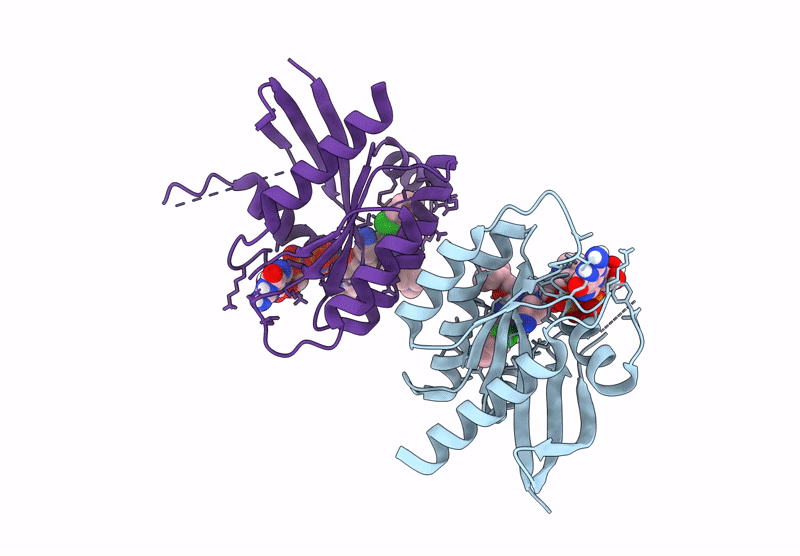
Crystal Structure of Kirsten Rat Sarcoma G12C Complexed with GMPPNP and Covalently Bound to an Adduct of {(2S)-4-[7-(8-chloronaphthalen-1-yl)-2-{[(2S)-1-methylpyrrolidin-2-yl]methoxy}-5,6,7,8-tetrahydropyrido[3,4-d]pyrimidin-4-yl]-1-[(2Z)-2-fluoro-3-(pyridin-2-yl)prop-2-enoyl]piperazin-2-yl}acetonitrile
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
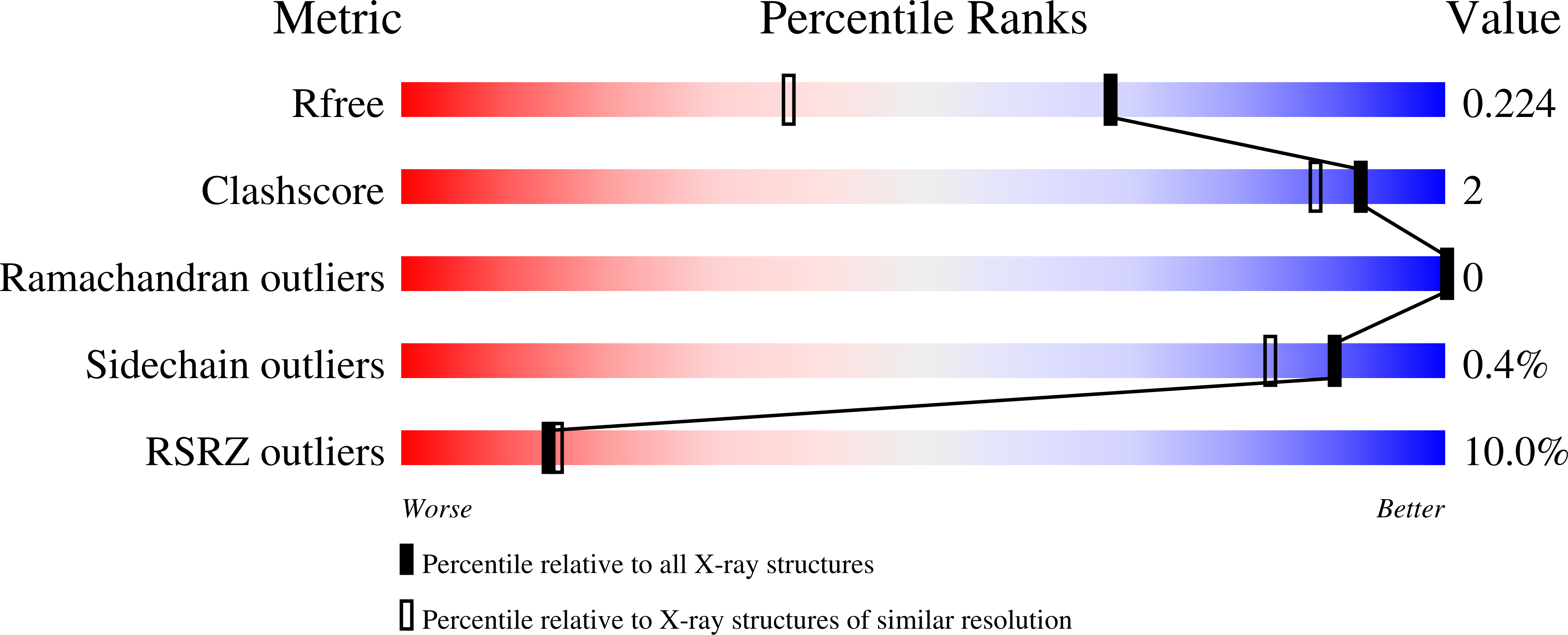
Resolution:
1.59 Å
R-Value Free:
0.22
R-Value Work:
0.20
R-Value Observed:
0.21
Space Group:
C 1 2 1