Deposition Date
2024-06-21
Release Date
2025-05-14
Last Version Date
2025-07-30
Entry Detail
PDB ID:
9IIU
Keywords:
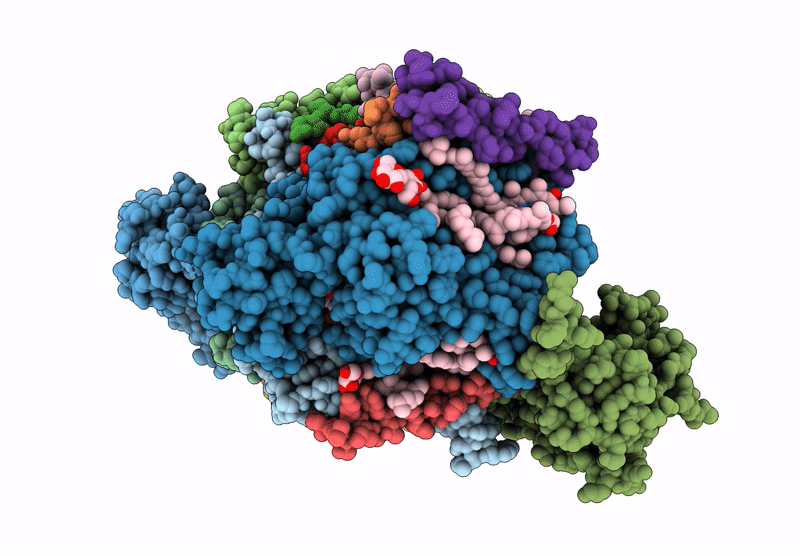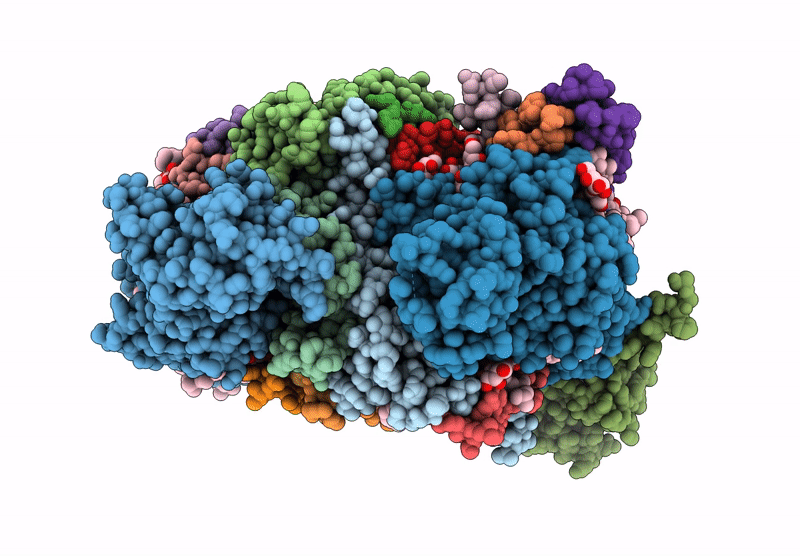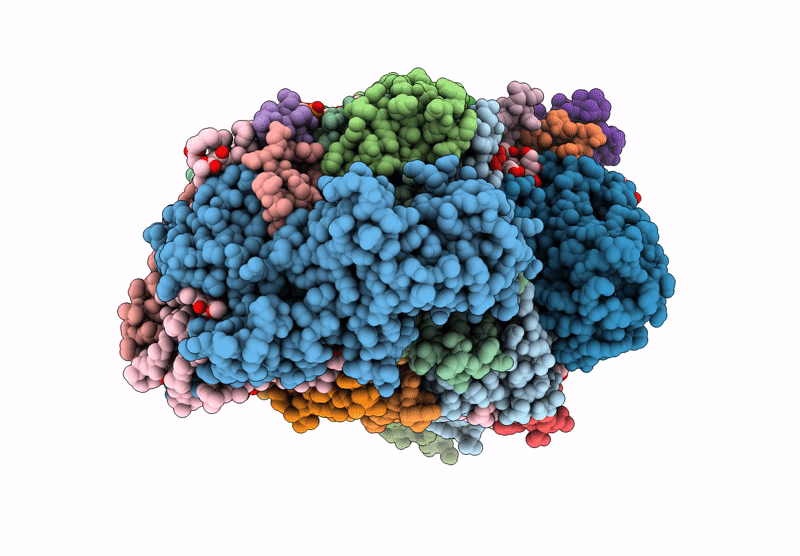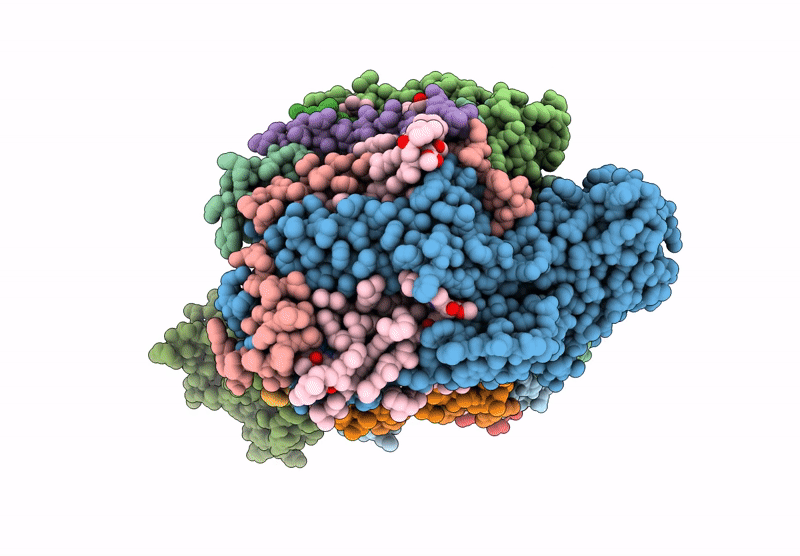
Title:
Cryo-EM structure of an TEF30-associated intermediate PSII core complex from Chlamydomonas reinhardtii
Biological Source:
Source Organism(s):
Chlamydomonas reinhardtii (Taxon ID: 3055)
Method Details:
Experimental Method:
Resolution:
2.98 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE


