Deposition Date
2025-01-24
Release Date
2025-03-12
Last Version Date
2025-04-30
Entry Detail
PDB ID:
9I4G
Keywords:
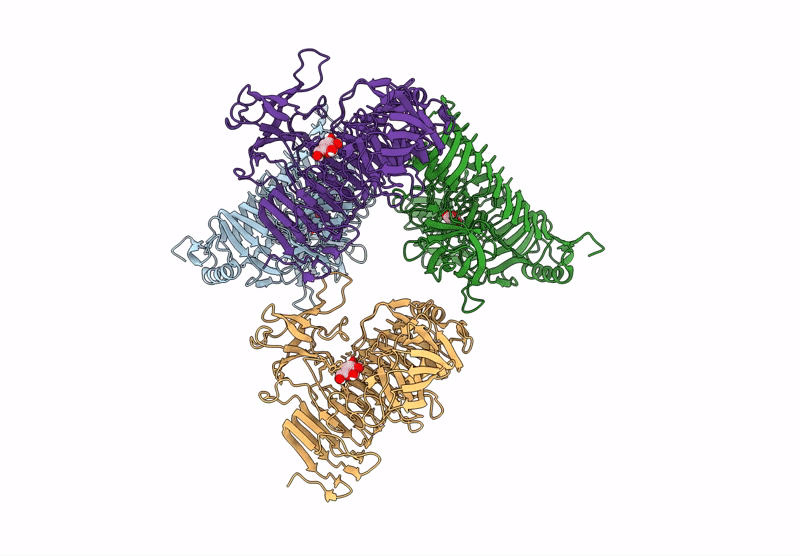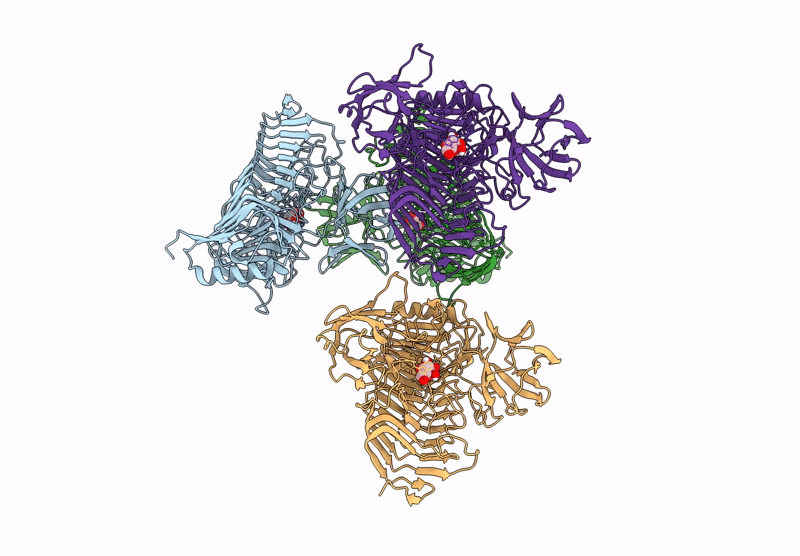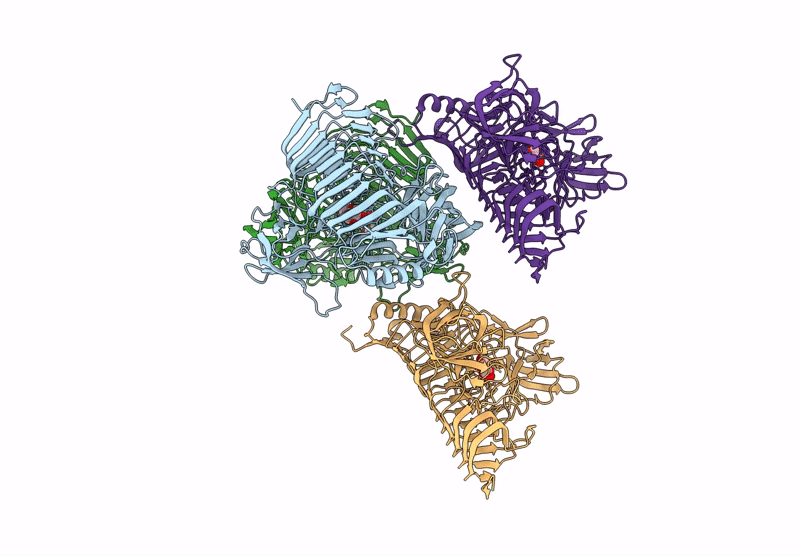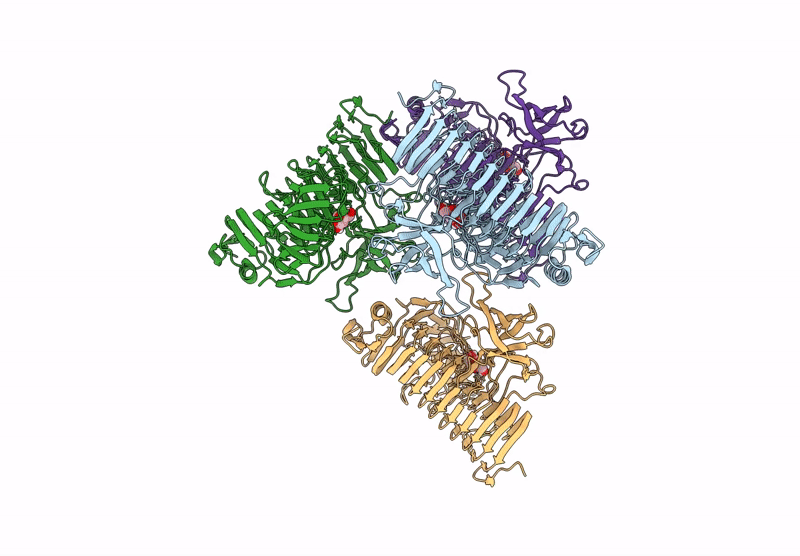
Title:
Blood Type B-converting alpha-1,3-galactosidase PpaGal from Pedobacter panaciterrae in complex with D-galactose
Biological Source:
Source Organism(s):
Pedobacter panaciterrae (Taxon ID: 363849)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.98 Å
R-Value Free:
0.26
R-Value Work:
0.22
Space Group:
P 1 21 1


