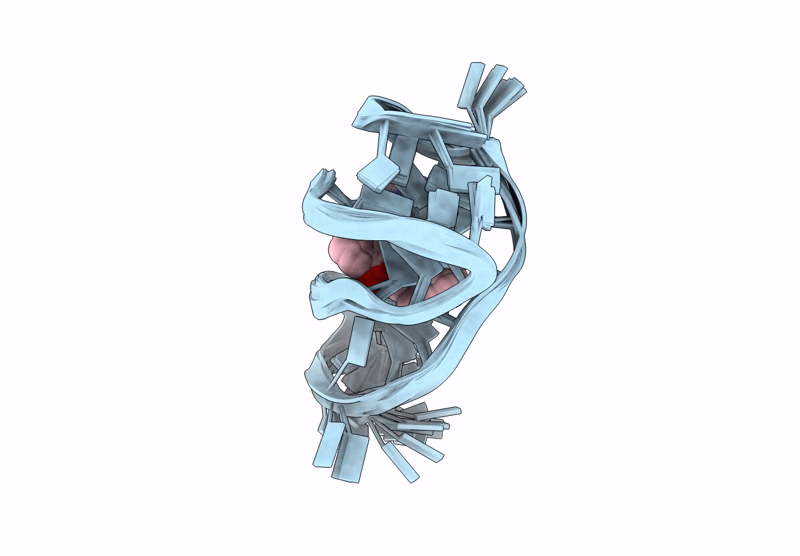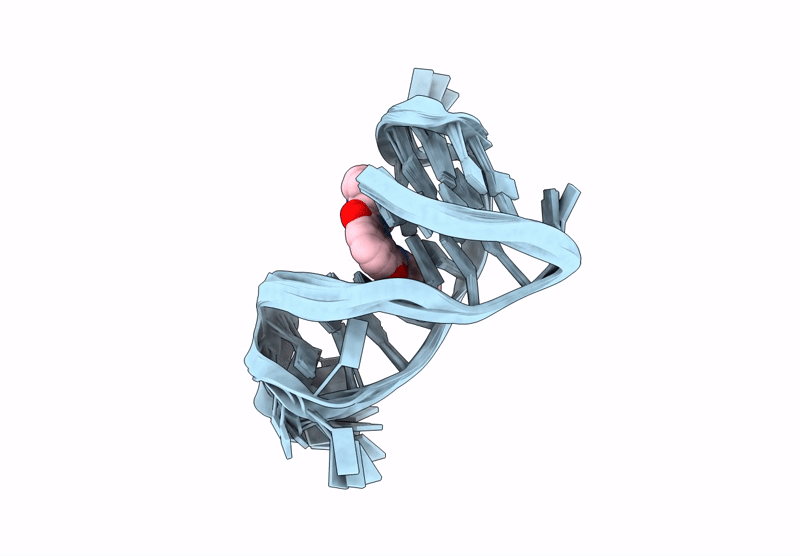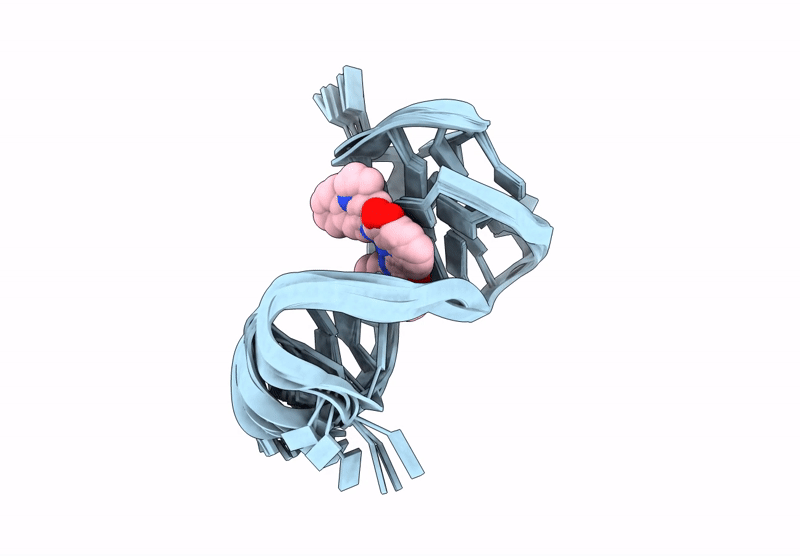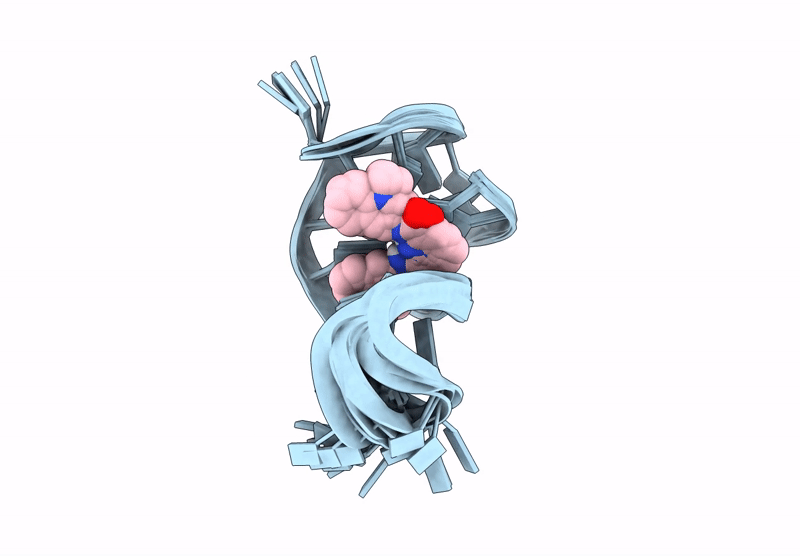
Deposition Date
2024-09-25
Release Date
2025-08-27
Last Version Date
2025-10-01
Entry Detail
PDB ID:
9GVV
Keywords:
Title:
Quadruplex-duplex hybrid (QDH) complex with 360A from PIM1 oncogene.
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Method Details:
Experimental Method:
Conformers Calculated:
200
Conformers Submitted:
10
Selection Criteria:
structures with the lowest energy