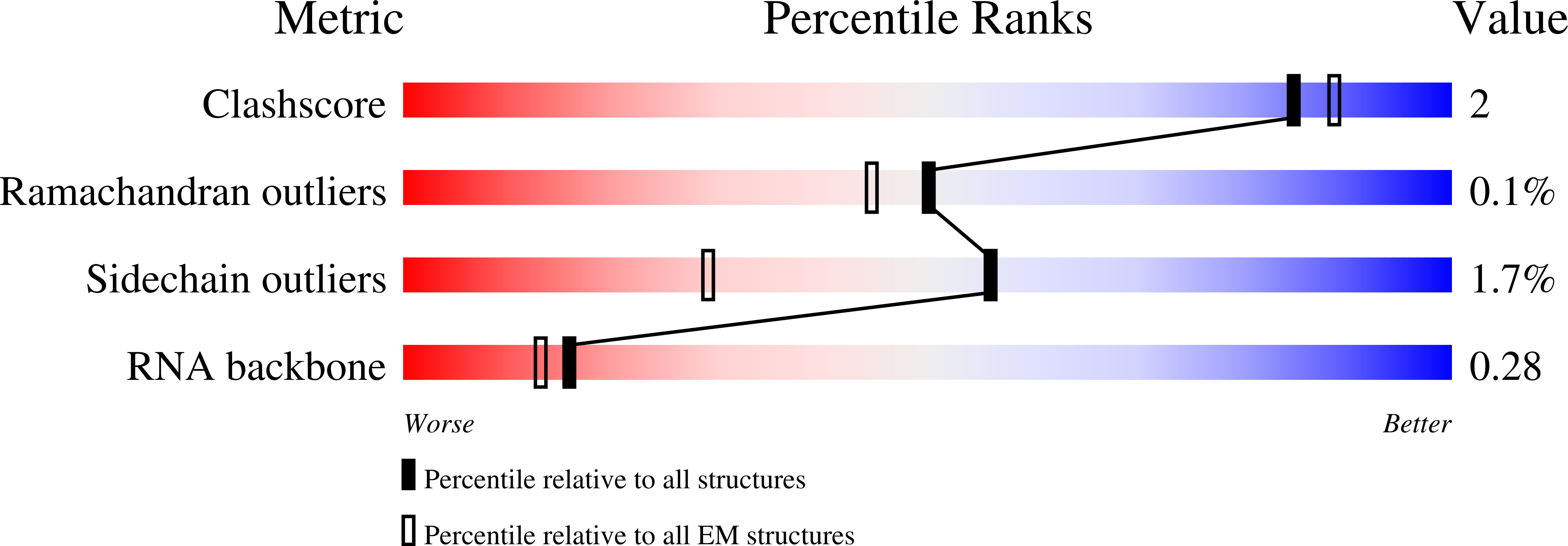
Deposition Date
2024-05-16
Release Date
2025-02-12
Last Version Date
2025-04-02
Entry Detail
PDB ID:
9FCV
Keywords:
Title:
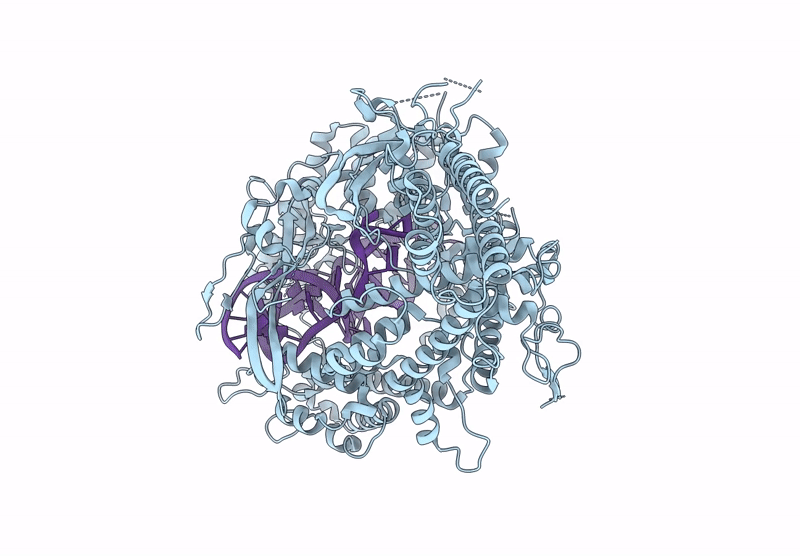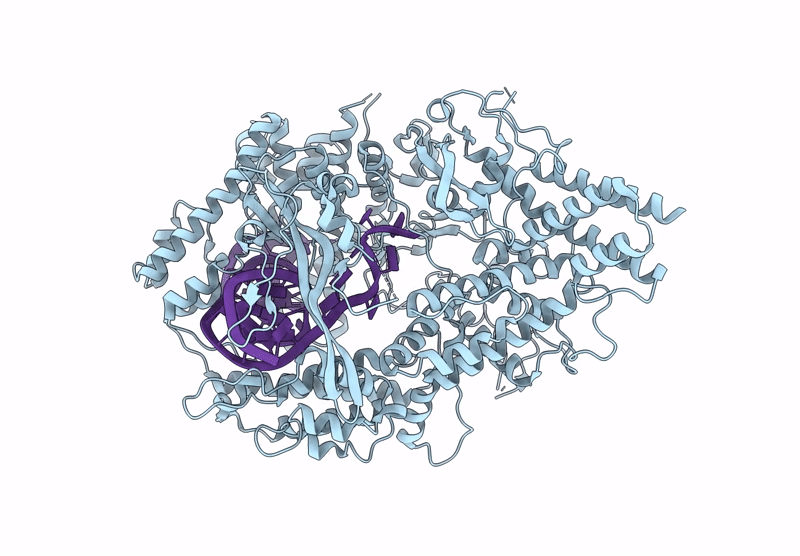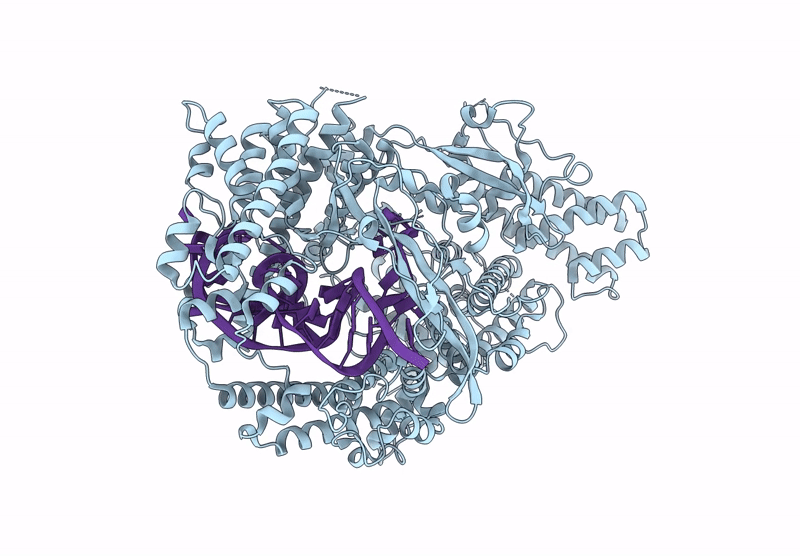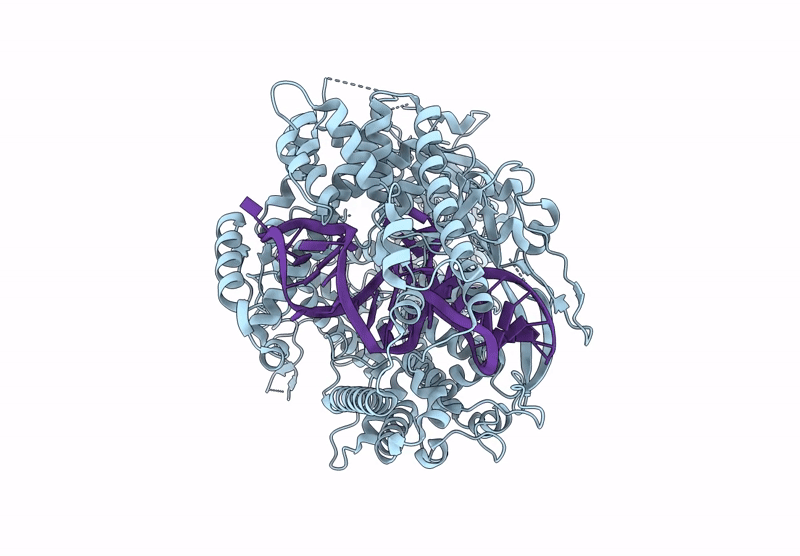
Cas nuclease-CRISPR (cr)RNA ribonucleoprotein (RNP) complex
Biological Source:
Source Organism(s):
Segatella buccae (Taxon ID: 28126)
synthetic construct (Taxon ID: 32630)
synthetic construct (Taxon ID: 32630)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.09 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE