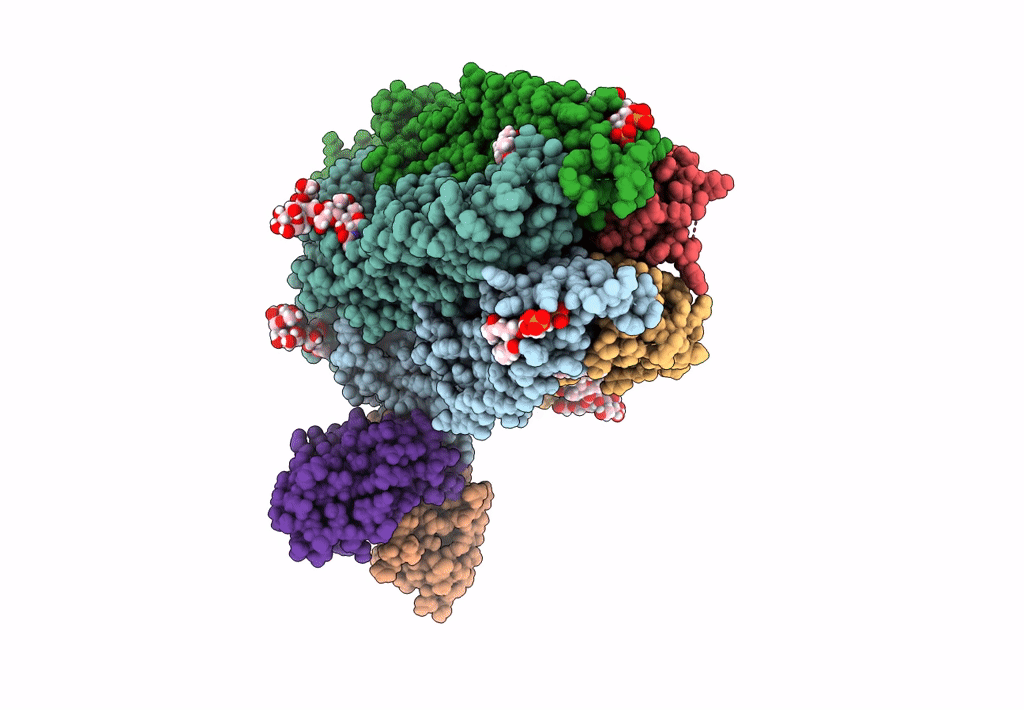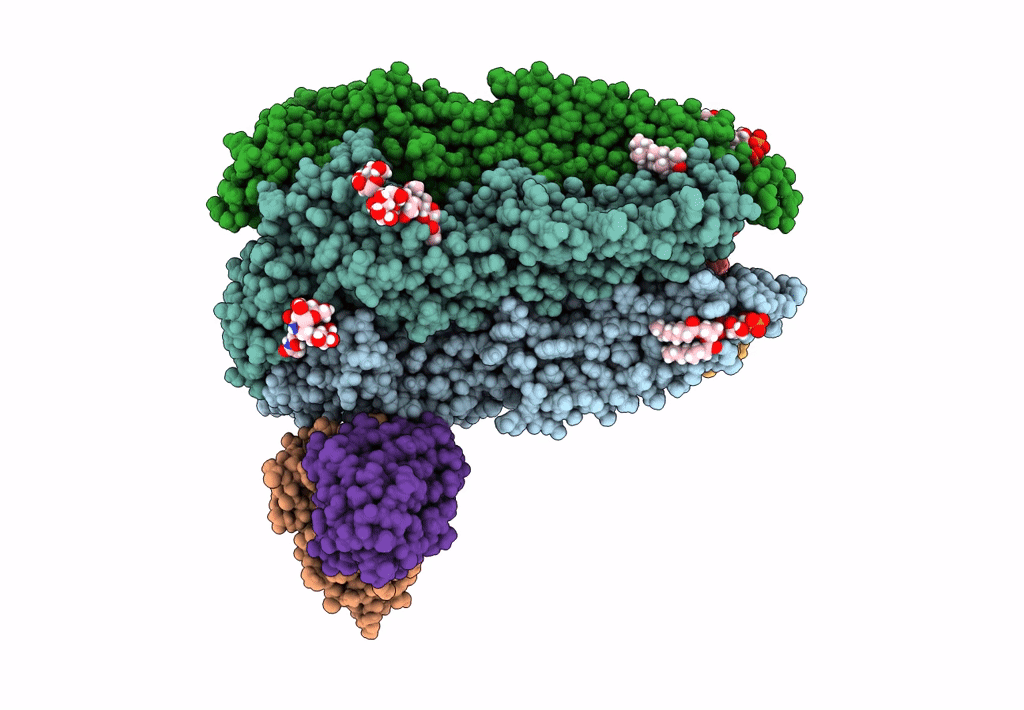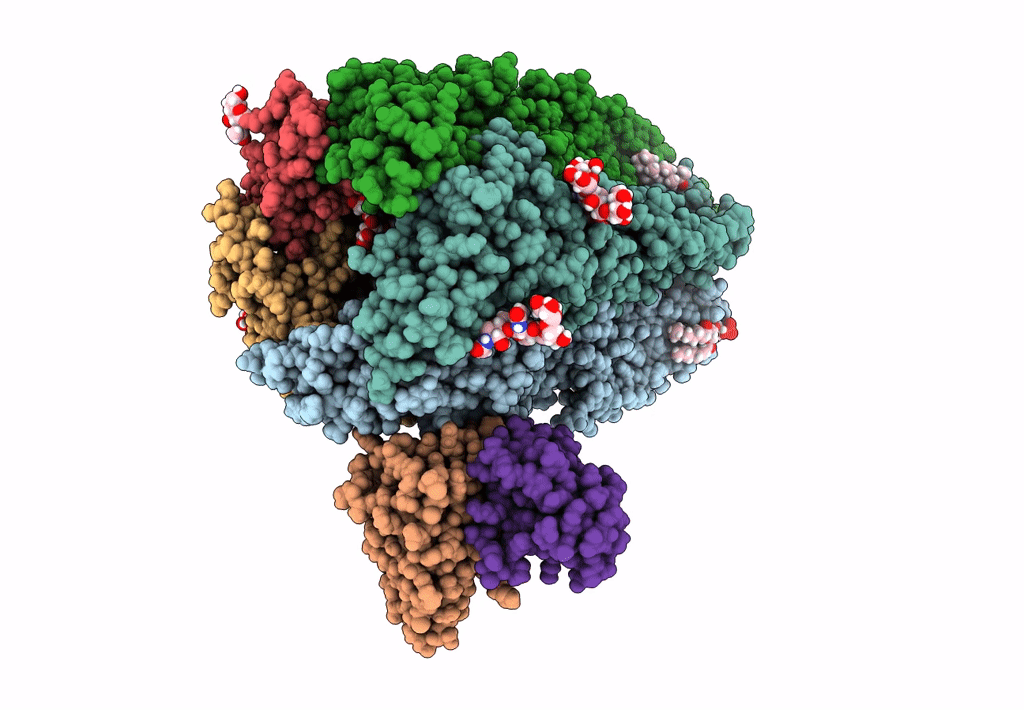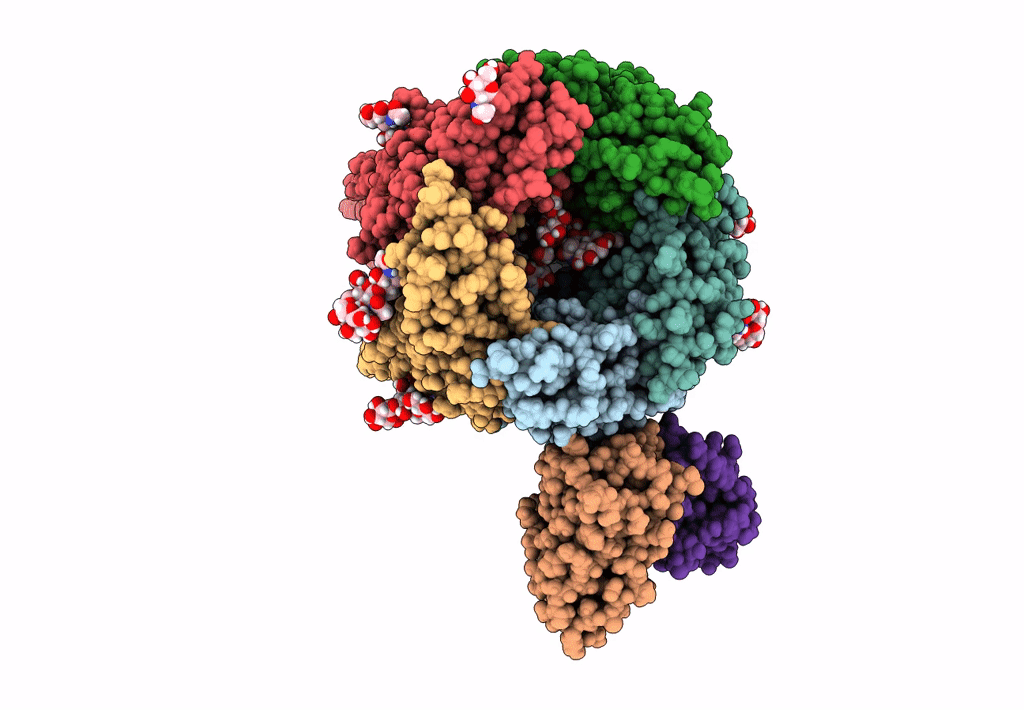
Deposition Date
2023-02-10
Release Date
2023-09-20
Last Version Date
2025-05-14
Entry Detail
PDB ID:
8G4X
Keywords:
Title:
Native GABA-A receptor from the mouse brain, meta-alpha1-alpha3-beta2-gamma2 subtype, in complex with GABA and allopregnanolone
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.56 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE


