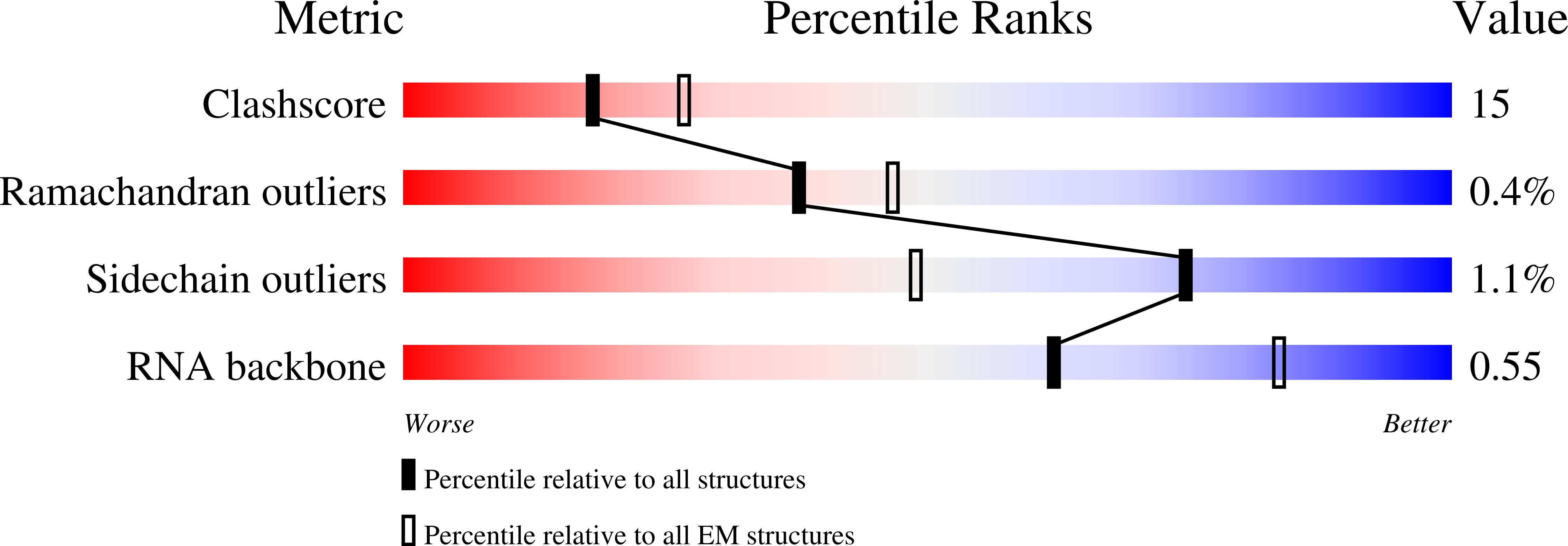Deposition Date
2022-12-18
Release Date
2023-03-29
Last Version Date
2024-06-19
Entry Detail
PDB ID:
8FIZ
Keywords:
Title:
Cryo-EM structure of E. coli 70S Ribosome containing mRNA and tRNA (in the transcription-translation complex)
Biological Source:
Source Organism(s):
Escherichia phage Lambda (Taxon ID: 2681611)
Escherichia coli (Taxon ID: 562)
Escherichia coli (Taxon ID: 562)
Method Details:
Experimental Method:
Resolution:
3.80 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE