Deposition Date
2022-07-21
Release Date
2023-08-02
Last Version Date
2023-11-29
Entry Detail
PDB ID:
8DS8
Keywords:
Title:
Crystal structure of human TNRC18 BAH domain in complex with H3K9me3 peptide
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
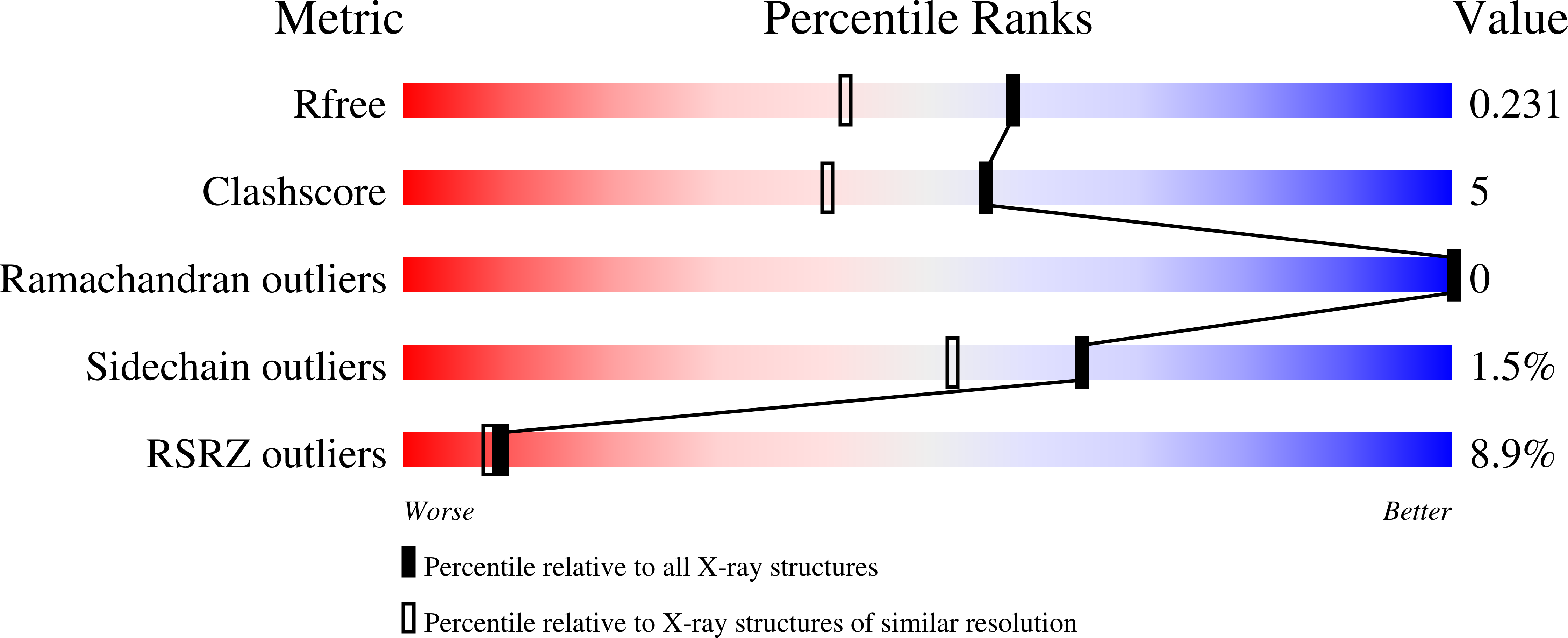
Resolution:
1.84 Å
R-Value Free:
0.23
R-Value Work:
0.20
R-Value Observed:
0.20
Space Group:
P 21 21 2