Deposition Date
2022-05-17
Release Date
2023-02-15
Last Version Date
2024-11-13
Entry Detail
PDB ID:
8CUZ
Keywords:
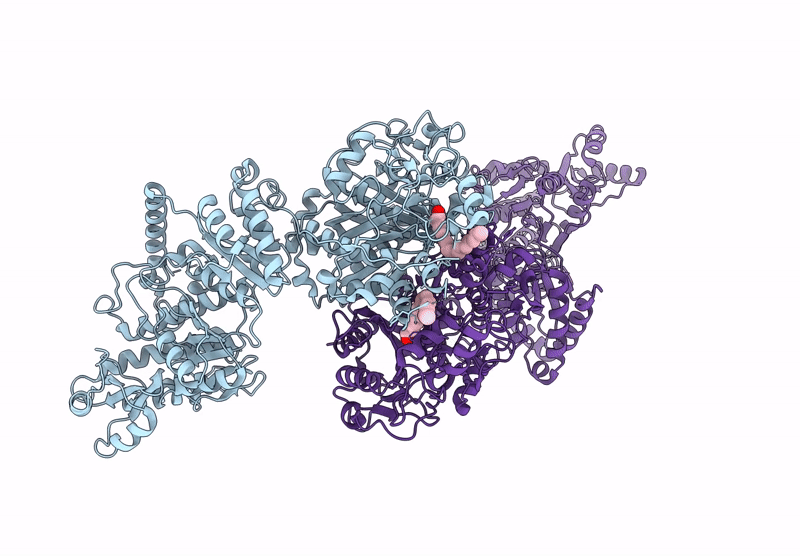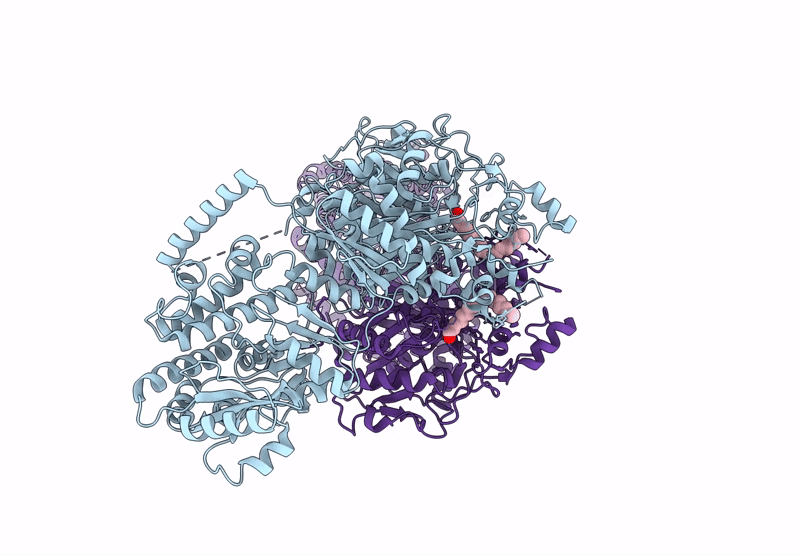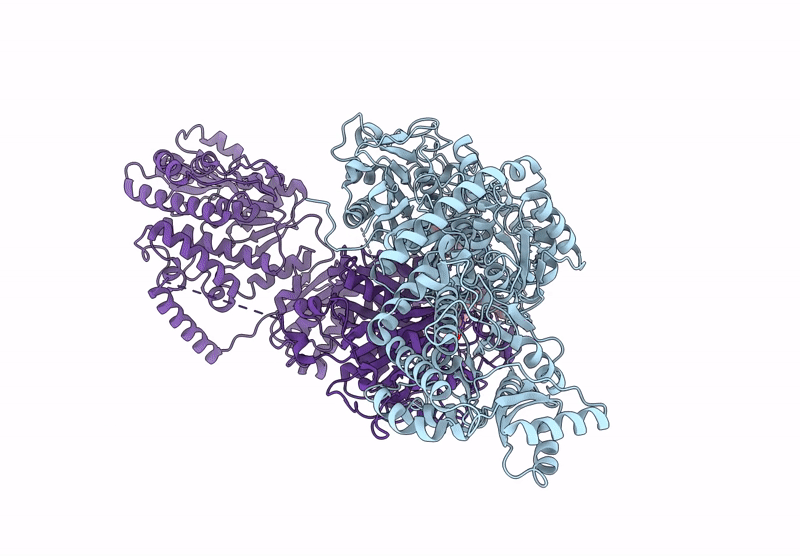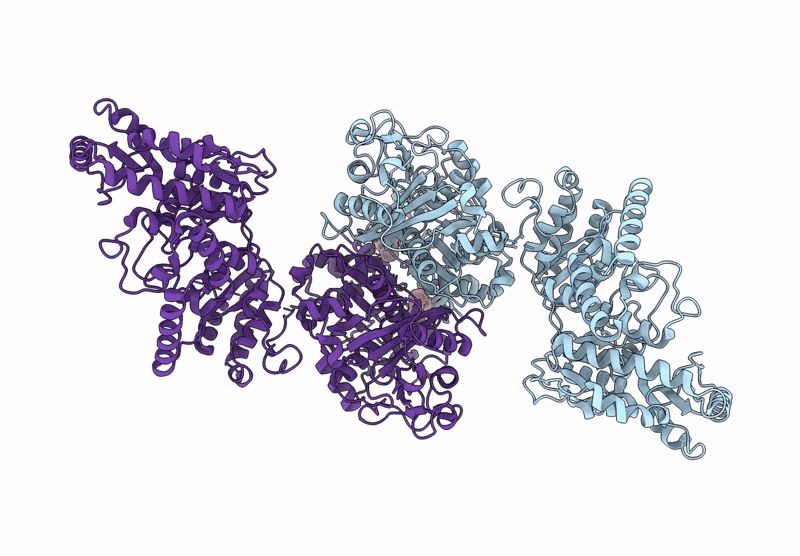
Title:
KS-AT domains of mycobacterial Pks13 with inward AT conformation
Biological Source:
Source Organism(s):
Mycolicibacterium smegmatis MC2 155 (Taxon ID: 246196)
Method Details:
Experimental Method:
Resolution:
3.00 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE


