Abstact
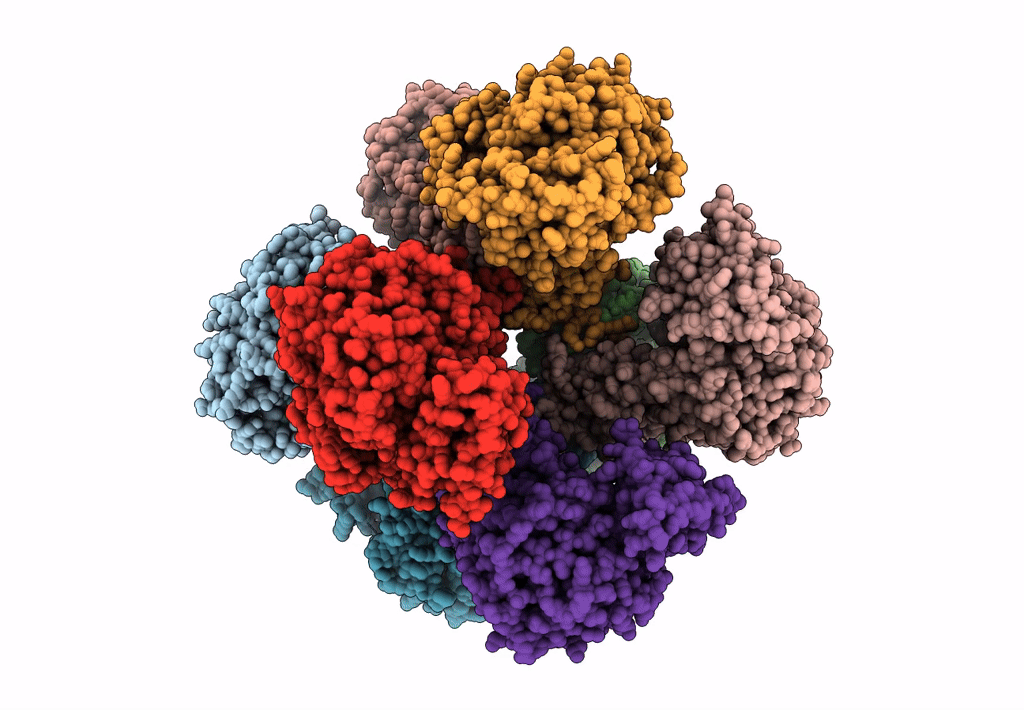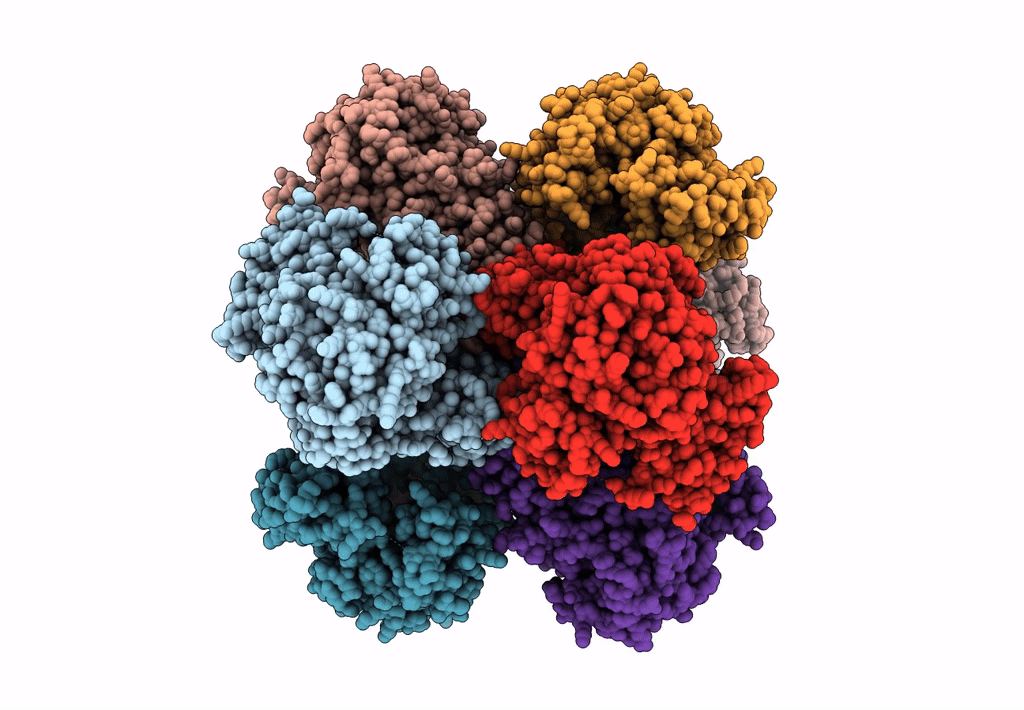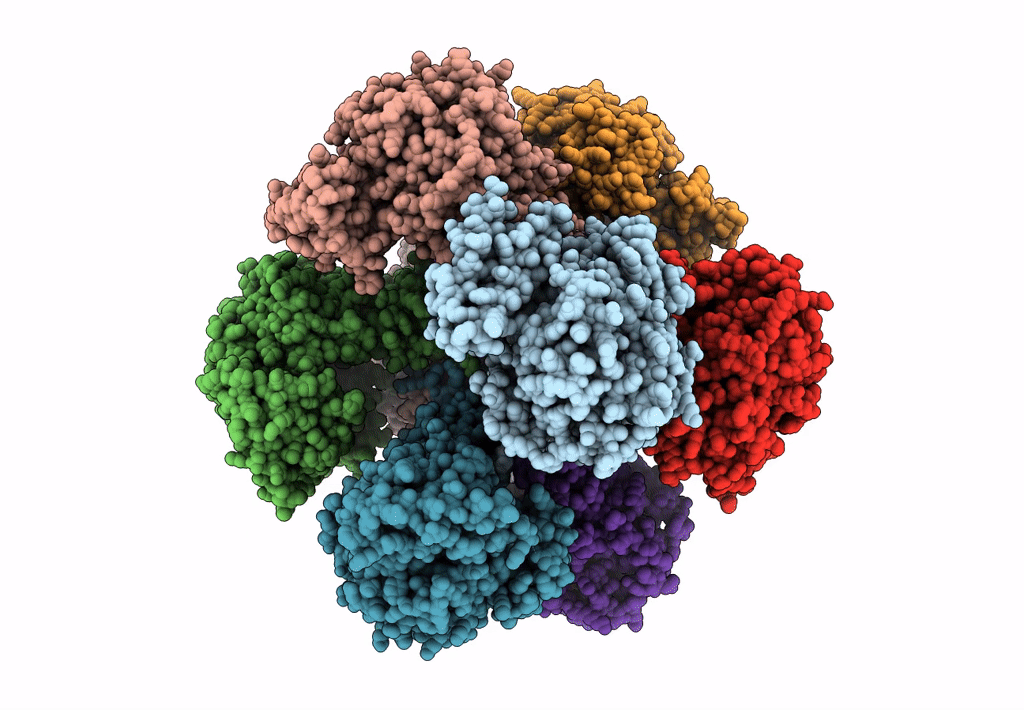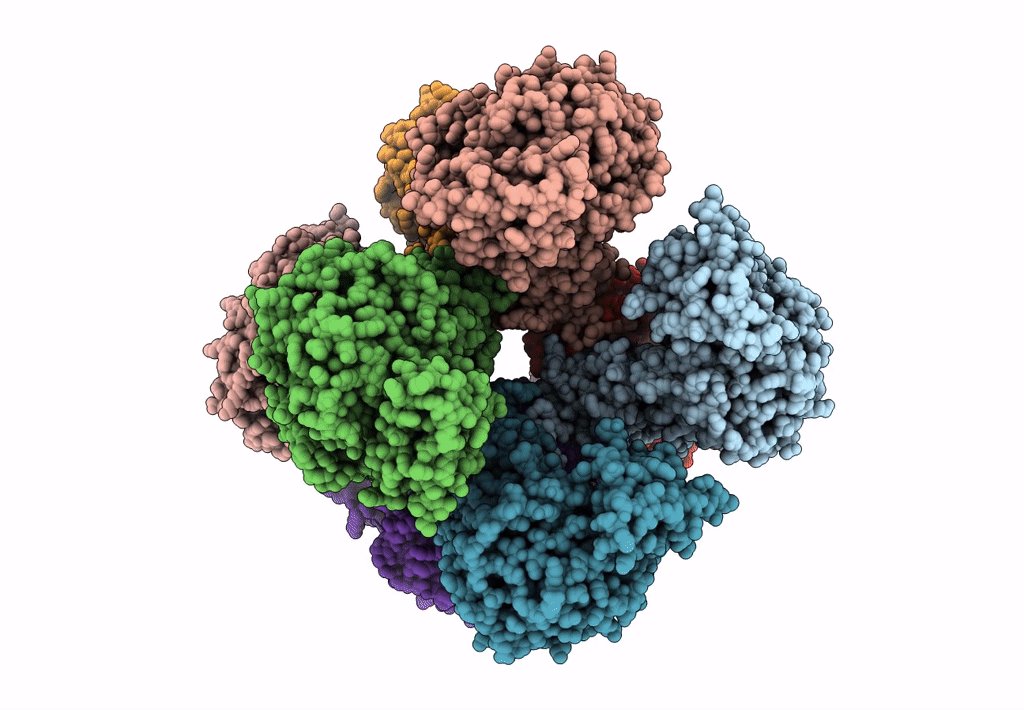
Invasive aspergillosis is one of the most serious clinical invasive fungal infections, resulting in a high case fatality rate among immunocompromised patients. The disease is caused by saprophytic molds in the genus Aspergillus, including Aspergillus fumigatus, the most significant pathogenic species. The fungal cell wall, an essential structure mainly composed of glucan, chitin, galactomannan, and galactosaminogalactan, represents an important target for the development of antifungal drugs. UDP (uridine diphosphate)-glucose pyrophosphorylase (UGP) is a central enzyme in the metabolism of carbohydrates that catalyzes the biosynthesis of UDP-glucose, a key precursor of fungal cell wall polysaccharides. Here, we demonstrate that the function of UGP is vital for Aspergillus nidulans (AnUGP). To understand the molecular basis of AnUGP function, we describe a cryoEM structure (global resolution of 3.5 Å for the locally refined subunit and 4 Å for the octameric complex) of a native AnUGP. The structure reveals an octameric architecture with each subunit comprising an N-terminal α-helical domain, a central catalytic glycosyltransferase A-like (GT-A-like) domain, and a C-terminal (CT) left-handed β-helix oligomerization domain. AnUGP displays unprecedented conformational variability between the CT oligomerization domain and the central GT-A-like catalytic domain. In combination with activity measurements and bioinformatics analysis, we unveil the molecular mechanism of substrate recognition and specificity for AnUGP. Altogether, our study not only contributes to understanding the molecular mechanism of catalysis/regulation of an important class of enzymes but also provides the genetic, biochemical, and structural groundwork for the future exploitation of UGP as a potential antifungal target. IMPORTANCE Fungi cause diverse diseases in humans, ranging from allergic syndromes to life-threatening invasive diseases, together affecting more than a billion people worldwide. Increasing drug resistance in Aspergillus species represents an emerging global health threat, making the design of antifungals with novel mechanisms of action a worldwide priority. The cryoEM structure of UDP (uridine diphosphate)-glucose pyrophosphorylase (UGP) from the filamentous fungus Aspergillus nidulans reveals an octameric architecture displaying unprecedented conformational variability between the C-terminal oligomerization domain and the central glycosyltransferase A-like catalytic domain in the individual protomers. While the active site and oligomerization interfaces are more highly conserved, these dynamic interfaces include motifs restricted to specific clades of filamentous fungi. Functional study of these motifs could lead to the definition of new targets for antifungals inhibiting UGP activity and, thus, the architecture of the cell wall of filamentous fungal pathogens.