Deposition Date
2022-07-05
Release Date
2022-09-28
Last Version Date
2024-01-31
Entry Detail
PDB ID:
8ACL
Keywords:
Title:
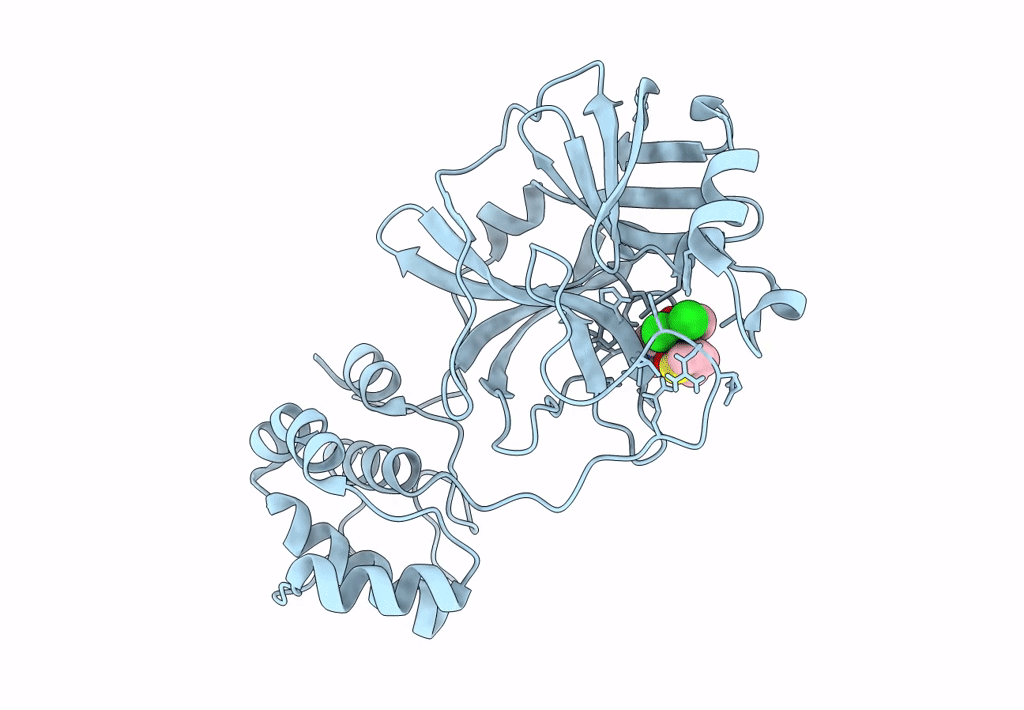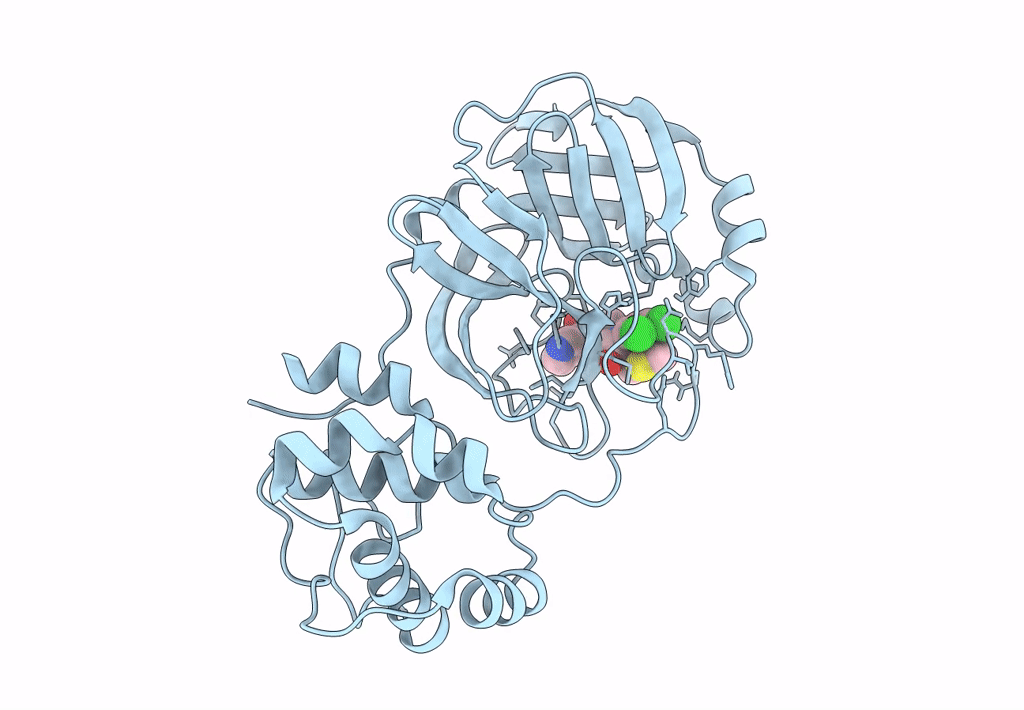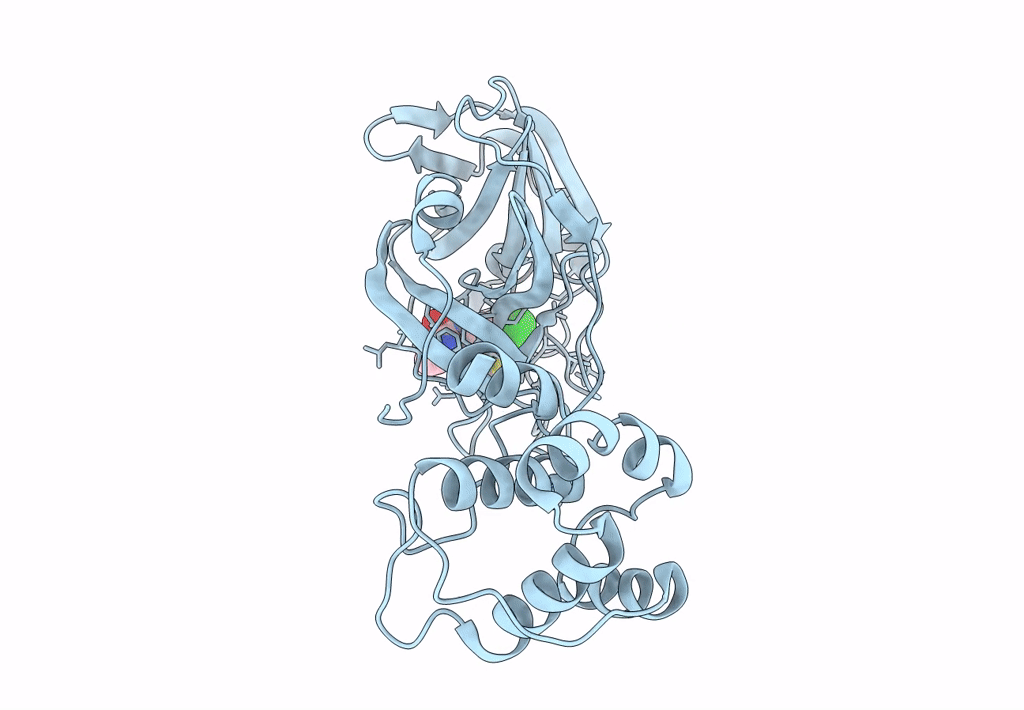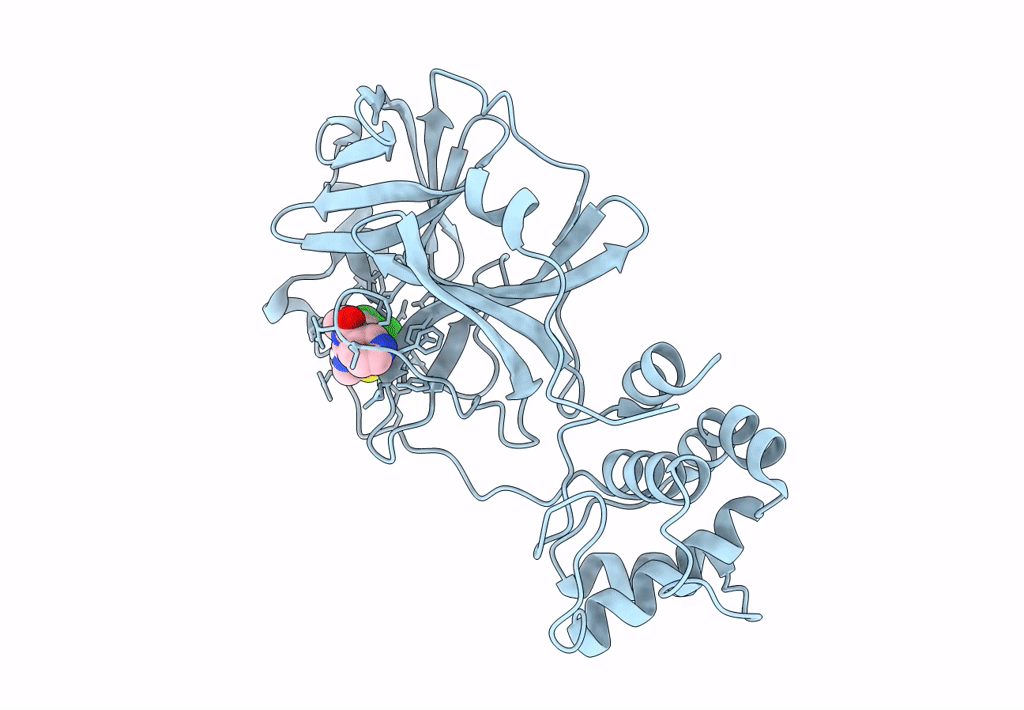
Crystal structure of SARS-CoV-2 main protease (MPro) in complex with the non-covalent inhibitor GC-14
Biological Source:
Source Organism(s):
Expression System(s):
Method Details:
Experimental Method:
Resolution:
1.40 Å
R-Value Free:
0.17
R-Value Work:
0.13
R-Value Observed:
0.13
Space Group:
C 1 2 1


