Deposition Date
2022-06-13
Release Date
2023-05-17
Last Version Date
2024-10-23
Entry Detail
PDB ID:
8A4O
Keywords:
Title:
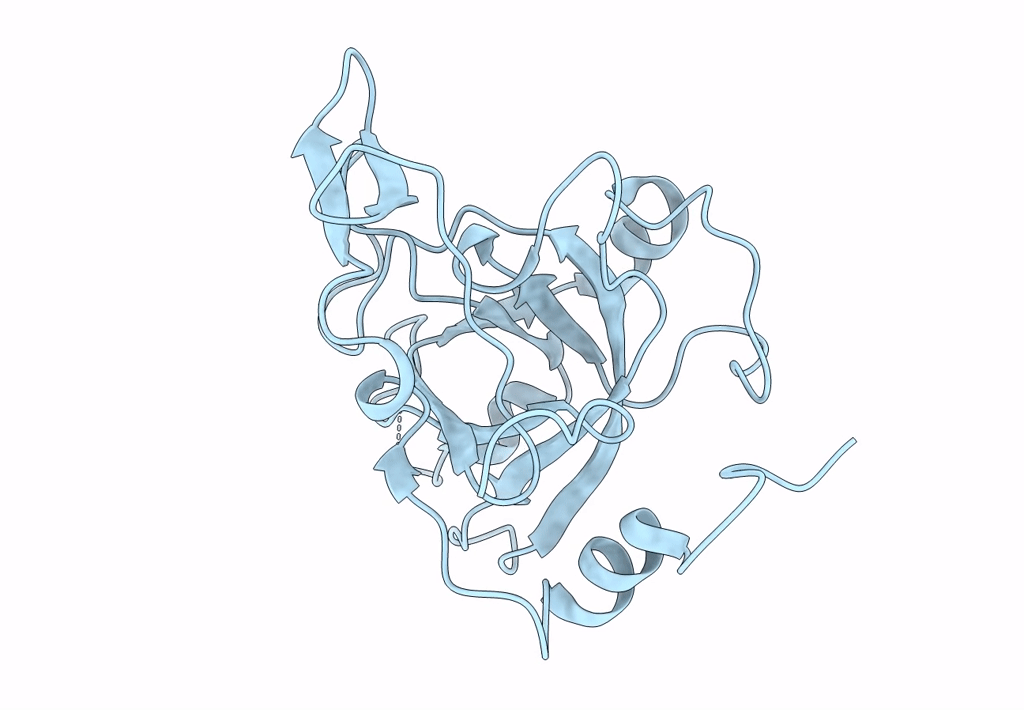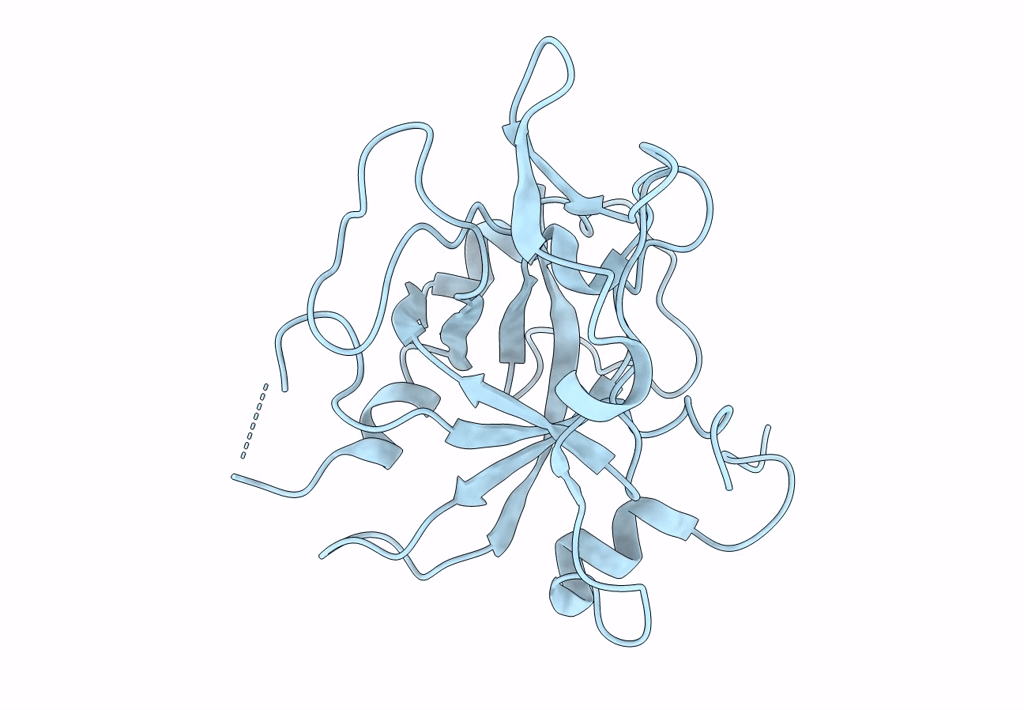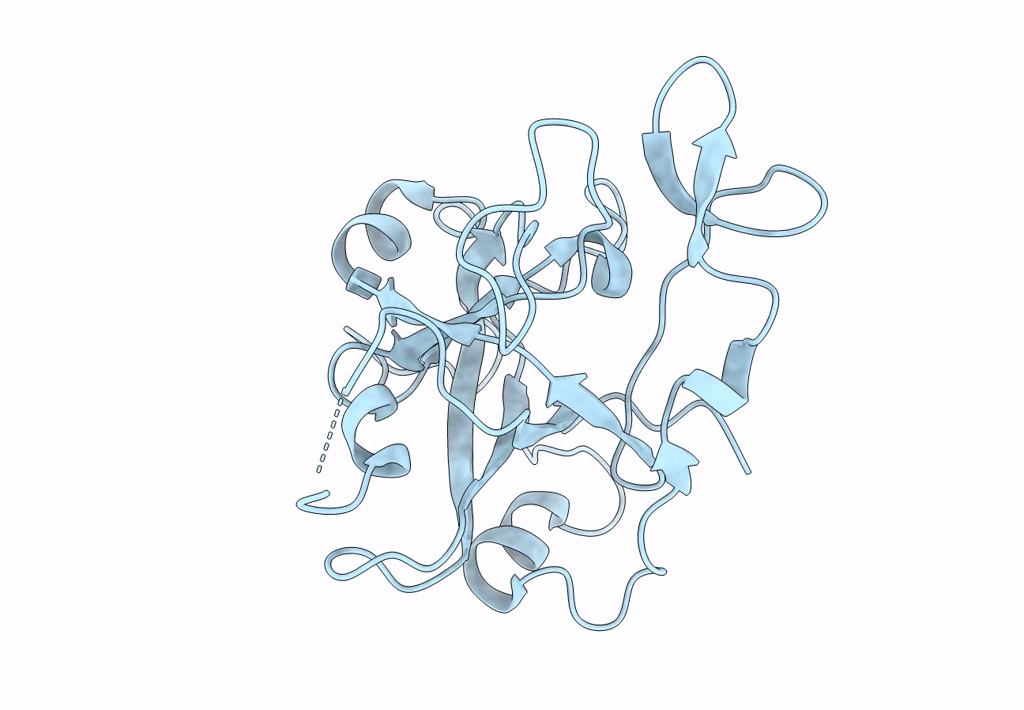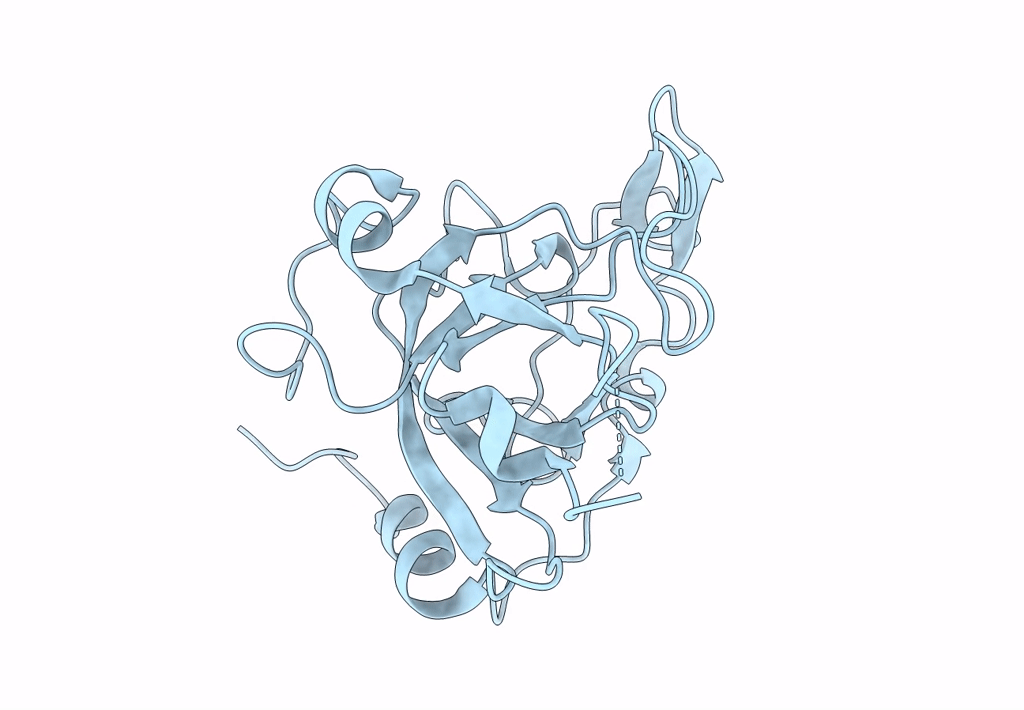
Crystal structure of the Ustilago hordei effector protein Uvi2
Biological Source:
Source Organism:
Ustilago hordei (Taxon ID: 120017)
Host Organism:
Method Details:
Experimental Method:
Resolution:
1.35 Å
R-Value Free:
0.18
R-Value Work:
0.16
R-Value Observed:
0.16
Space Group:
P 61 2 2


