Deposition Date
2021-08-10
Release Date
2022-04-06
Last Version Date
2023-11-29
Entry Detail
PDB ID:
7V2V
Keywords:
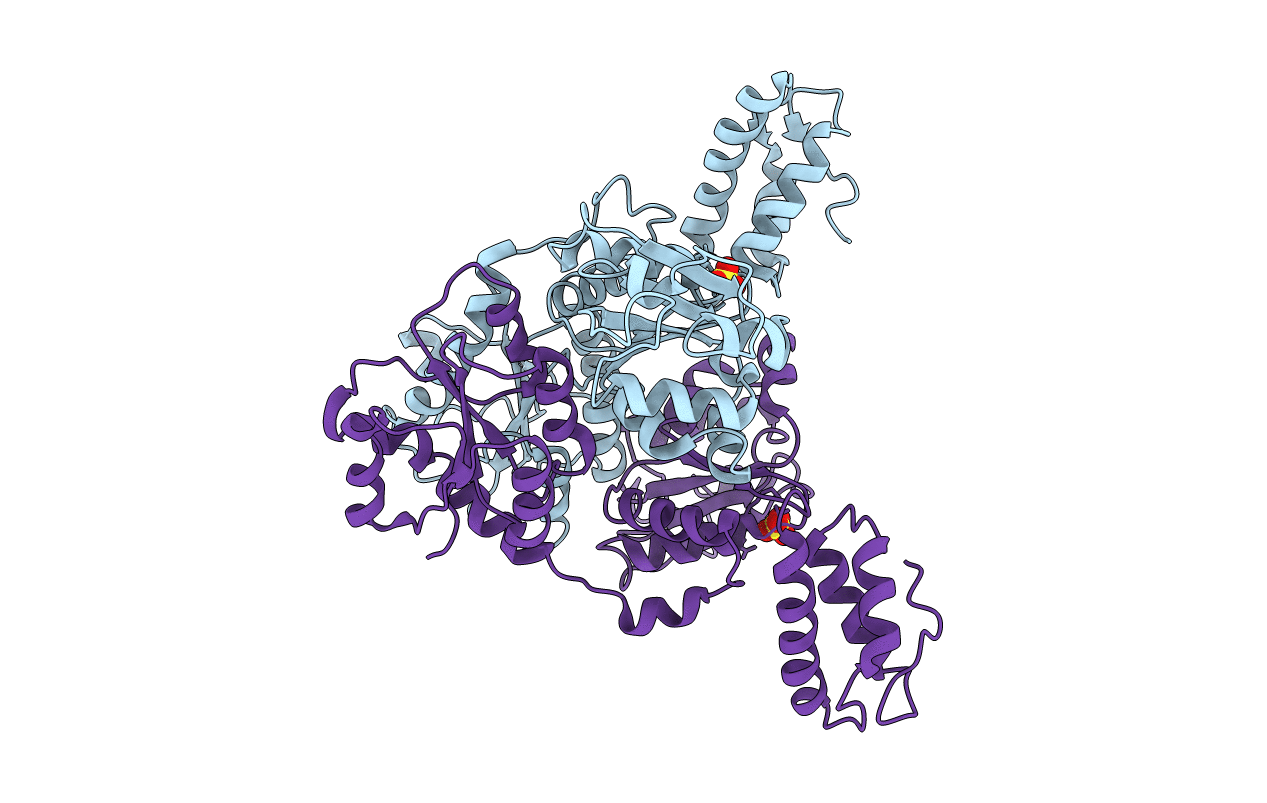
Title:
Crystal Structure of VpsR display novel dimeric architecture and c-di-GMP binding: mechanistic implications in oligomerization, ATPase activity and DNA binding.
Biological Source:
Source Organism(s):
Vibrio cholerae (Taxon ID: 666)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.19 Å
R-Value Free:
0.26
R-Value Work:
0.23
R-Value Observed:
0.23
Space Group:
P 31


