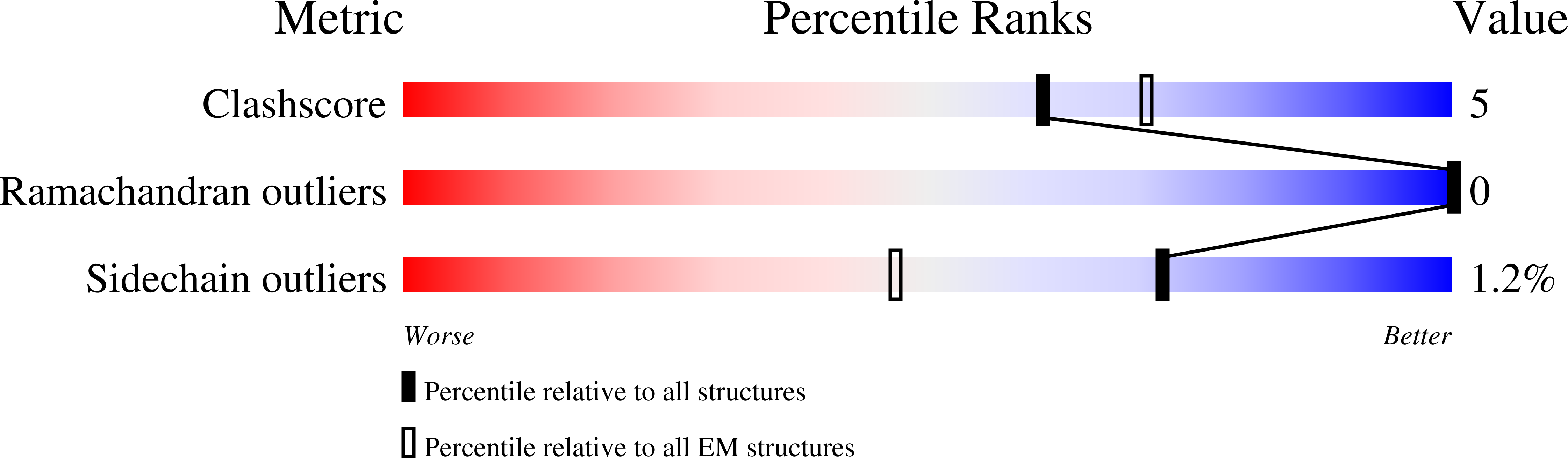
Deposition Date
2022-04-21
Release Date
2022-09-07
Last Version Date
2024-11-13
Entry Detail
PDB ID:
7URG
Keywords:
Title:
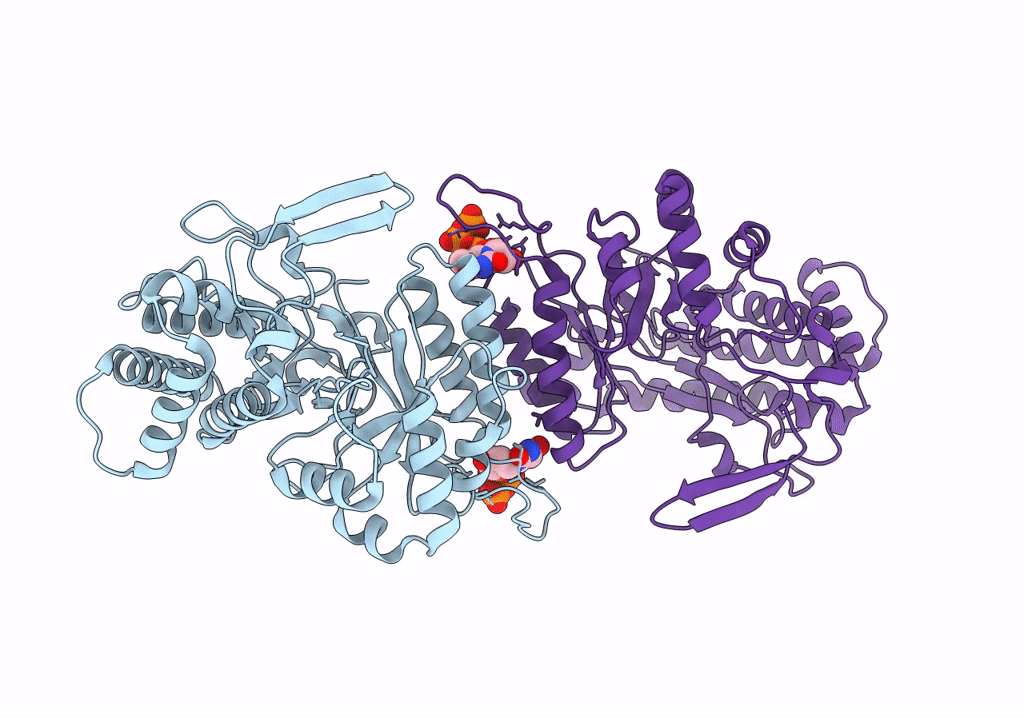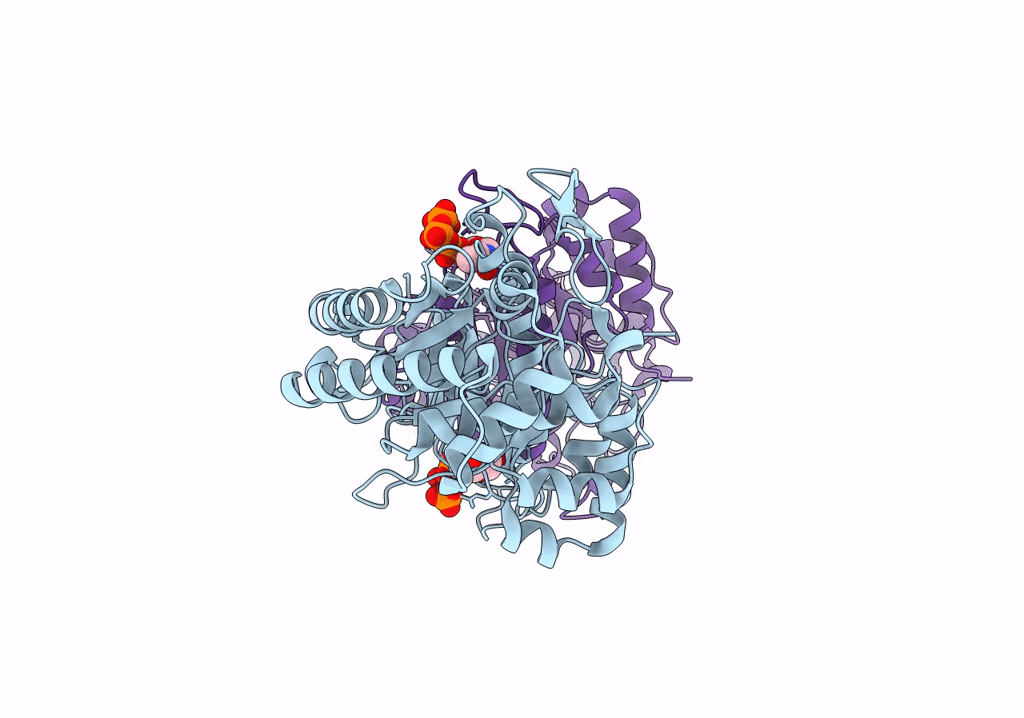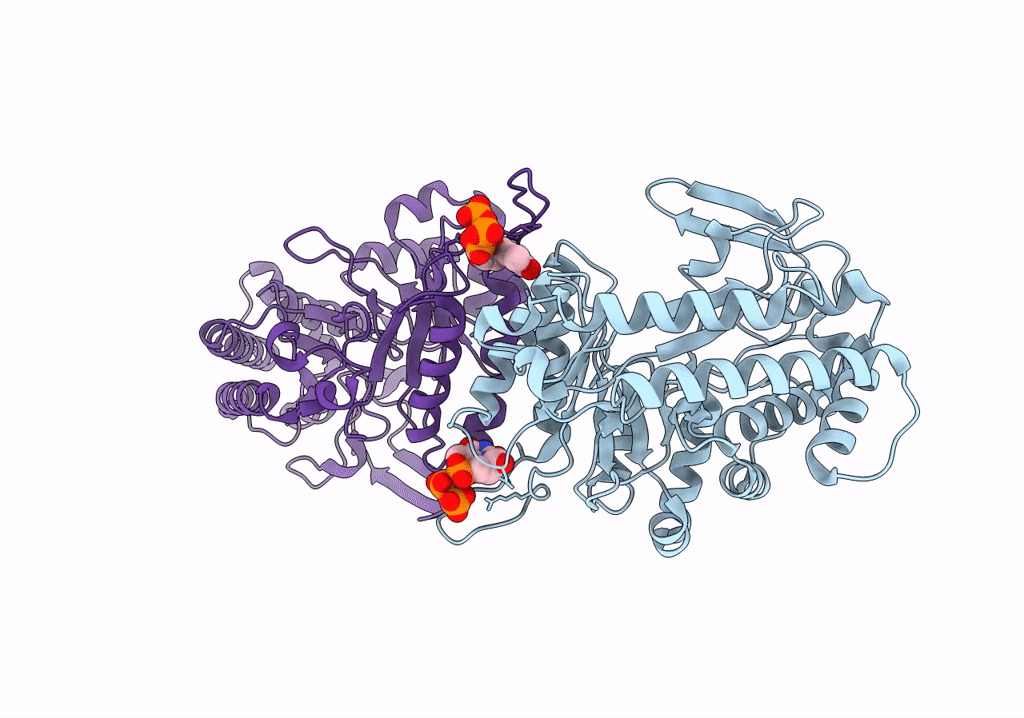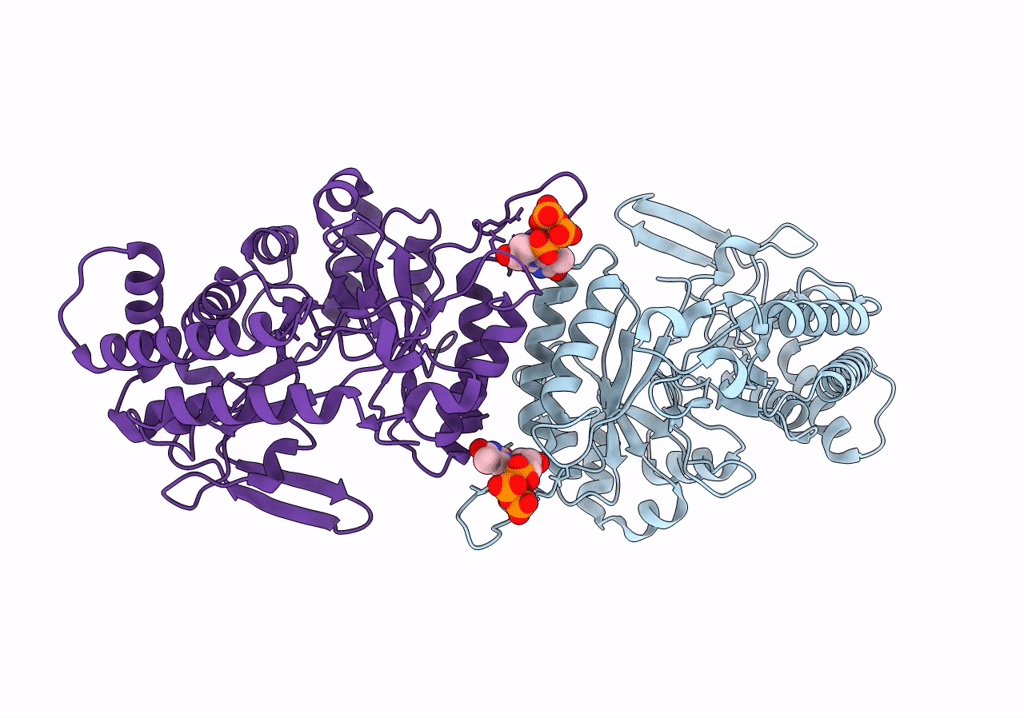
cryo-EM structure of ribonucleotide reductase from Synechococcus phage S-CBP4 bound with TTP
Biological Source:
Source Organism(s):
Synechococcus phage S-CBP4 (Taxon ID: 754059)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.46 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE