Abstact
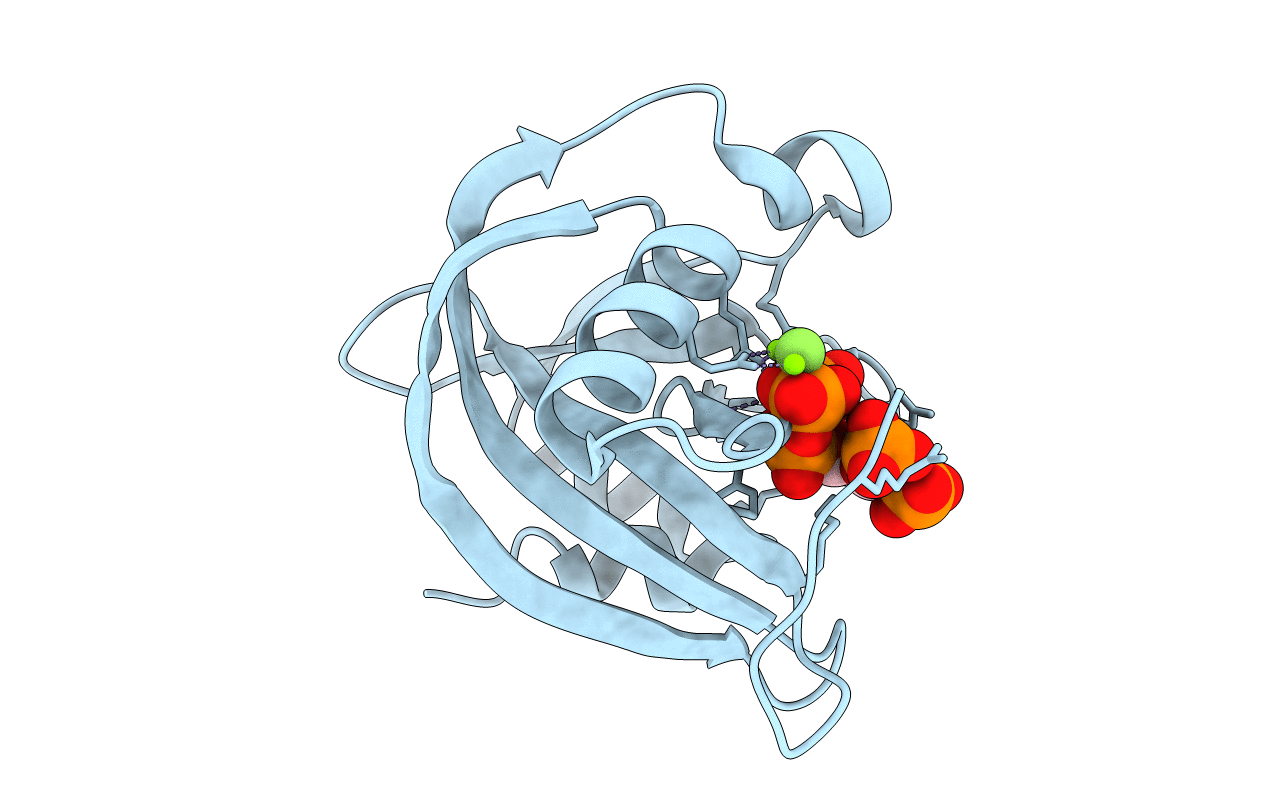
Inositol phosphate signaling in plants is of substantial agricultural interest, with a considerable focus on the inositol tris/tetrakisphosphate kinase (ITPK) family of inositol phosphate kinases. Historically, the 4-6 isoforms of ITPKs that higher plants each express have been studied for their multiplexing a metabolic pathway to synthesize inositol hexakisphosphate (ie InsP6 or phytate), through the phosphorylation and dephosphorylation of multiple inositol phosphates, including Ins(1,3,4,5,6)P5 (inositol-1,3,4,5,6-pentakisphosphate). A more recent discovery is ITPK-catalyzed phosphorylation of InsP6 to inositol pyrophosphates, which regulate plant immunity and phosphate homeostasis. However, a molecular-based explanation for these alternate catalytic activities has been missing, because no plant ITPK structure has previously been solved. Herein, we provide biochemical and structural analyses of ITPKs from Zea mays and Glycine max. For this work we introduce a simple, enzyme-coupled microplate-based assay of InsP6 kinase activity that should promote more general access to this important field. Furthermore, a ZmITPK1/InsP6 crystal complex is described at a resolution of 2.6 Å, which identifies a number of catalytically important residues; their functionality is confirmed by mutagenesis. We further demonstrate that ZmITPK1 adds a β-phosphate to the 3-position of Ins(1,2,3,4,5)P5 , yielding a candidate signal for regulating phosphate homeostasis. An impactful discovery is our description of a 29-residue catalytic specificity element; by interchanging this element between GmITPK1 and GmITPK2, we demonstrate how its isoform-specific sequence specifically determines whether the host protein phosphorylates InsP6 , without substantially affecting Ins(1,3,4,5,6)P5 metabolism. Our structural rationalization of key catalytic differences between alternate ITPK isoforms will complement future research into their functional diversity.