Deposition Date
2021-07-13
Release Date
2021-09-22
Last Version Date
2024-11-06
Entry Detail
PDB ID:
7FC3
Keywords:
Title:
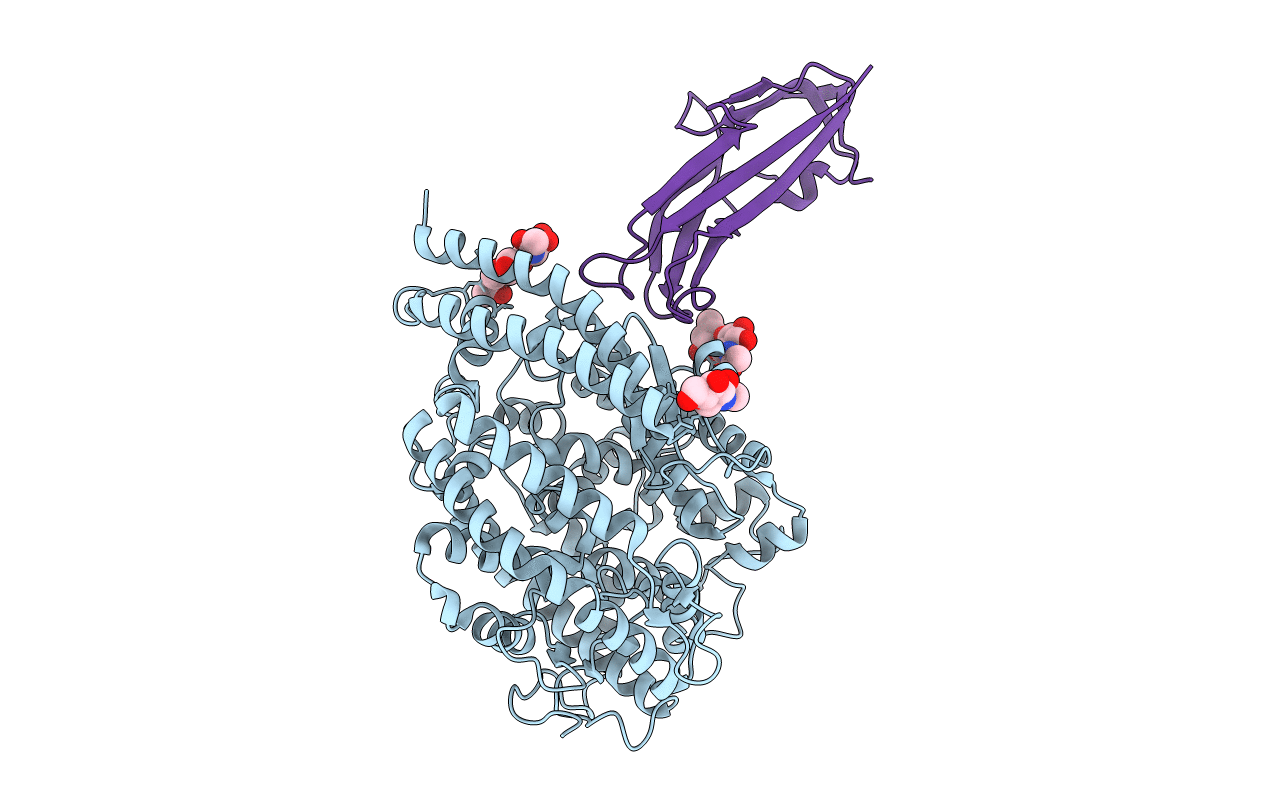
structure of NL63 receptor-binding domain complexed with horse ACE2
Biological Source:
Source Organism(s):
Human coronavirus NL63 (Taxon ID: 277944)
Equus caballus (Taxon ID: 9796)
Equus caballus (Taxon ID: 9796)
Expression System(s):
Method Details:
Experimental Method:
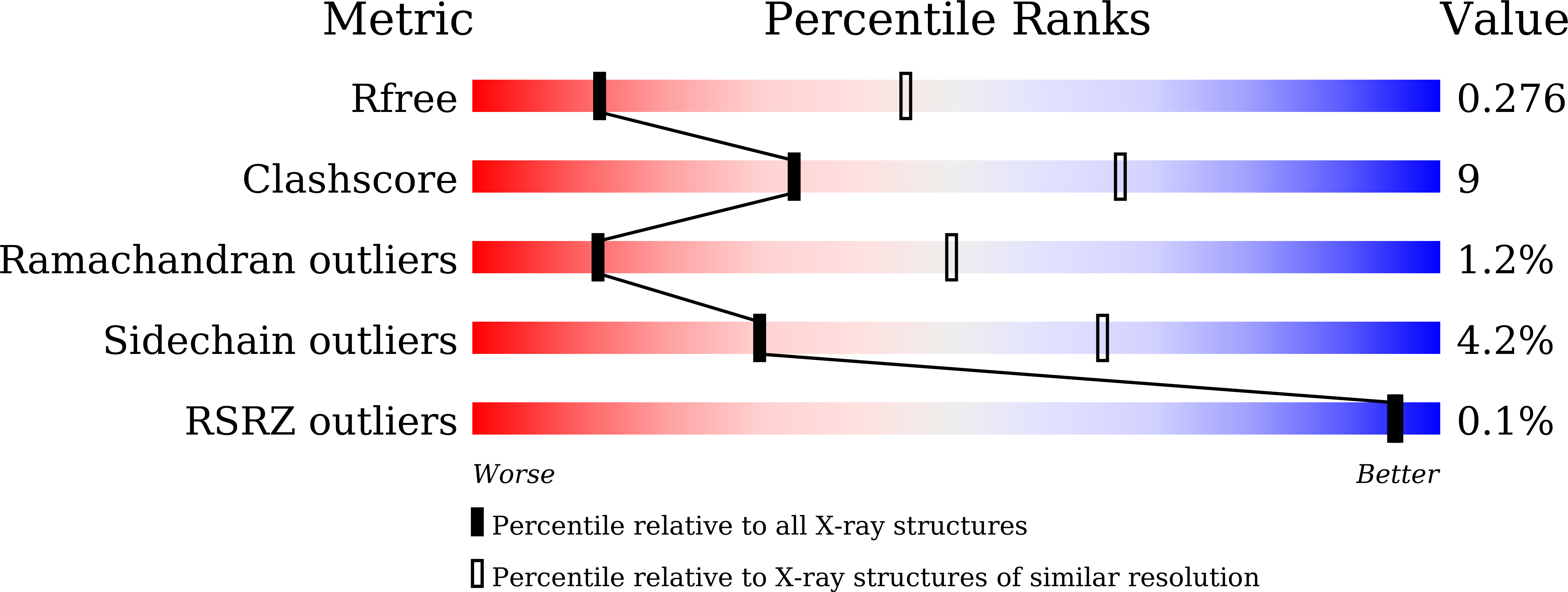
Resolution:
3.19 Å
R-Value Free:
0.28
R-Value Work:
0.23
R-Value Observed:
0.23
Space Group:
P 61 2 2