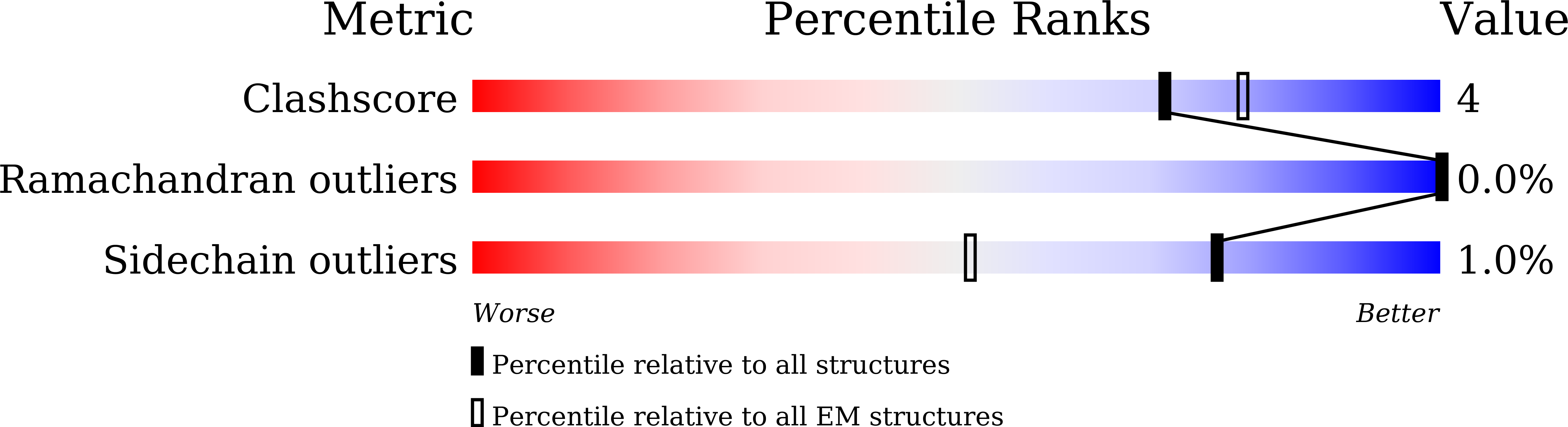
Deposition Date
2021-04-15
Release Date
2021-12-22
Last Version Date
2023-07-05
Entry Detail
PDB ID:
7EMY
Keywords:
Title:
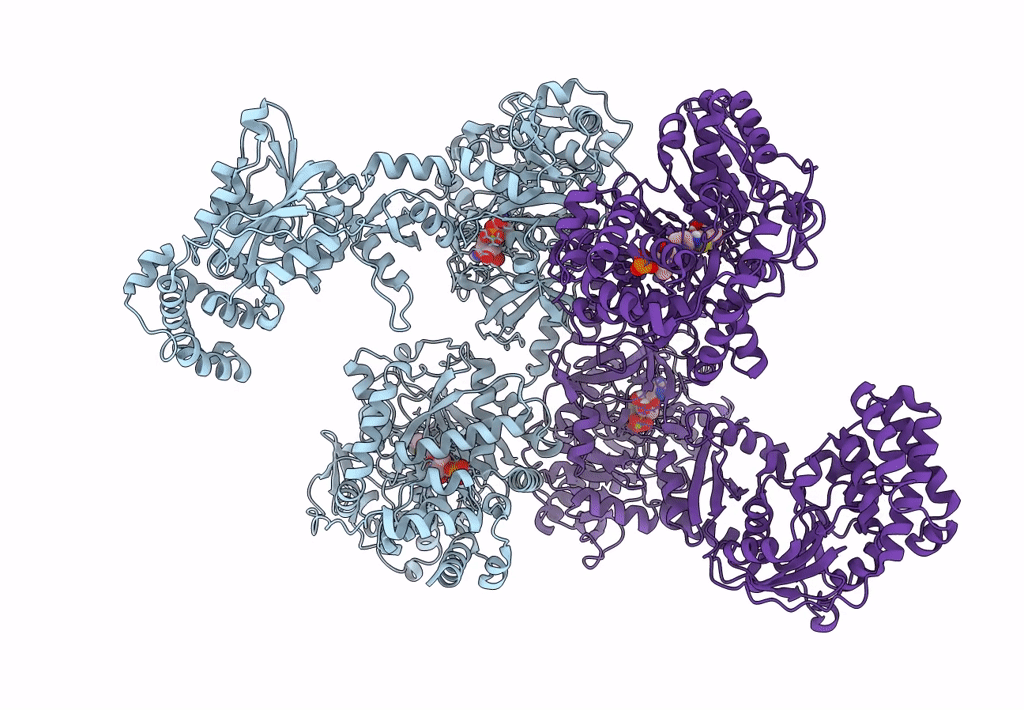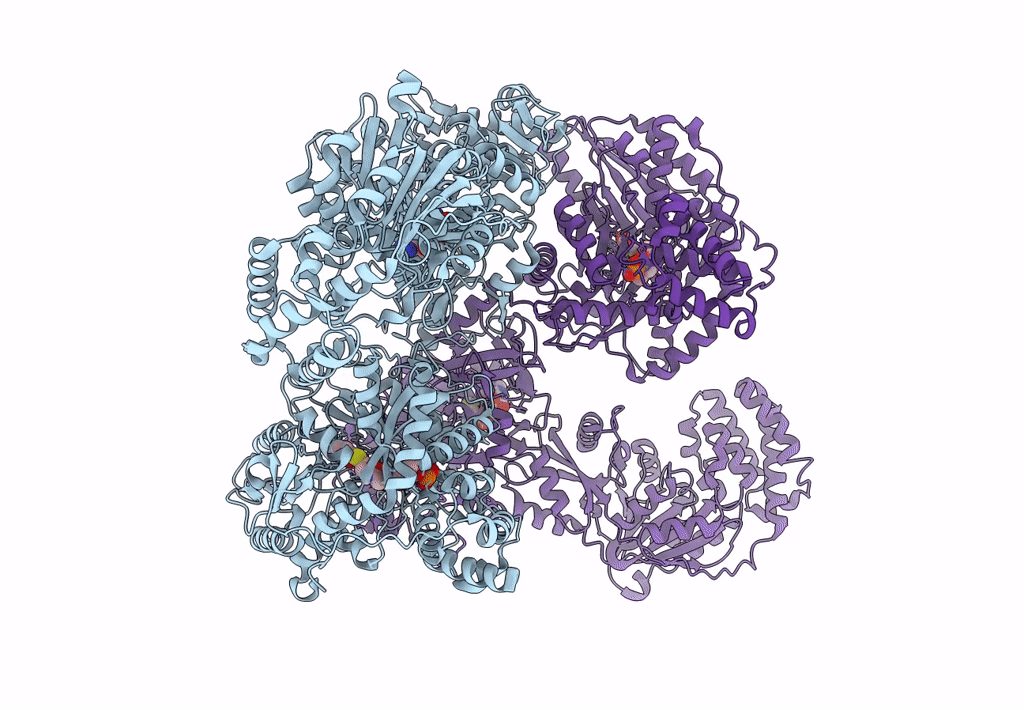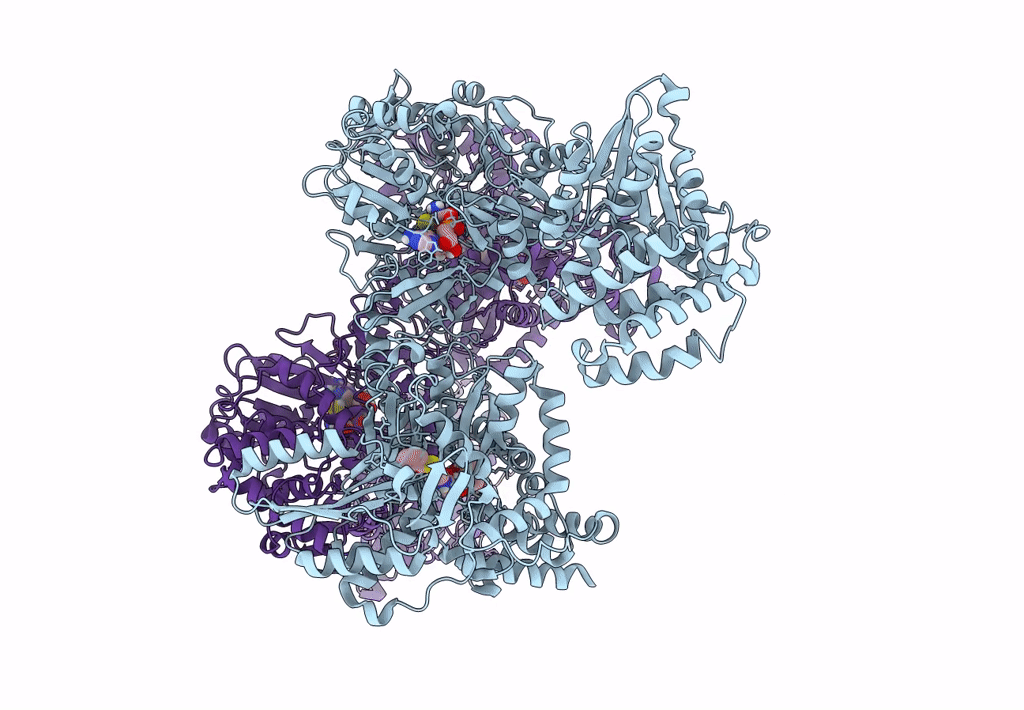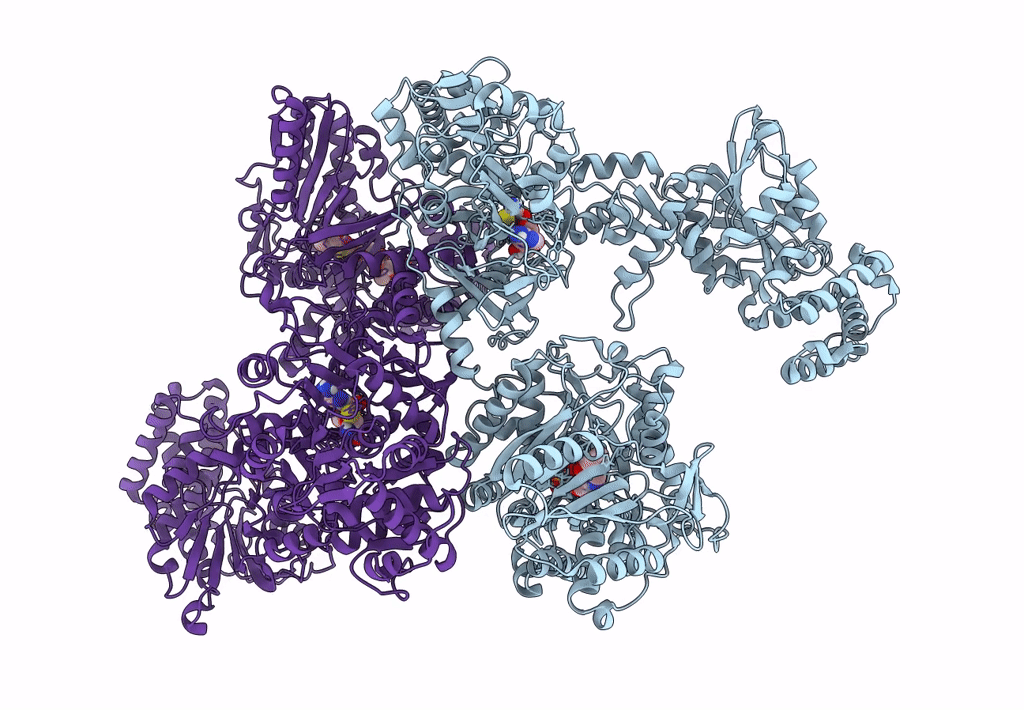
Pyochelin synthetase, a dimeric nonribosomal peptide synthetase elongation module
Biological Source:
Source Organism(s):
Pseudomonas aeruginosa PAO1 (Taxon ID: 208964)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.97 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE