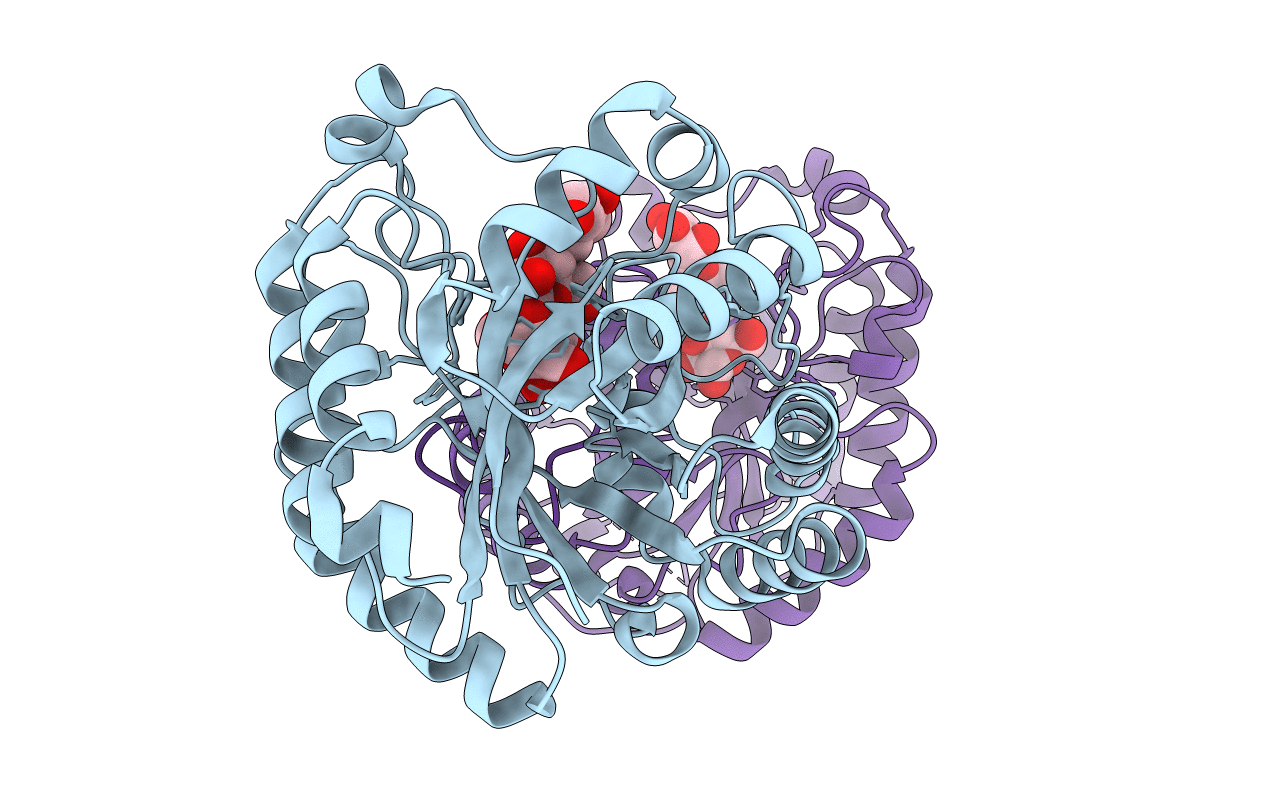
Deposition Date
2021-01-15
Release Date
2021-06-02
Last Version Date
2023-11-29
Entry Detail
PDB ID:
7DWA
Keywords:
Title:
Structure of a novel beta-mannanase BaMan113A with mannotriose, N236Y mutation
Biological Source:
Source Organism(s):
Bacillus sp. N16-5 (Taxon ID: 122631)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
1.62 Å
R-Value Free:
0.17
R-Value Work:
0.14
R-Value Observed:
0.14
Space Group:
P 2 21 21


