Deposition Date
2019-09-28
Release Date
2019-12-04
Last Version Date
2024-10-16
Entry Detail
PDB ID:
6UHV
Keywords:
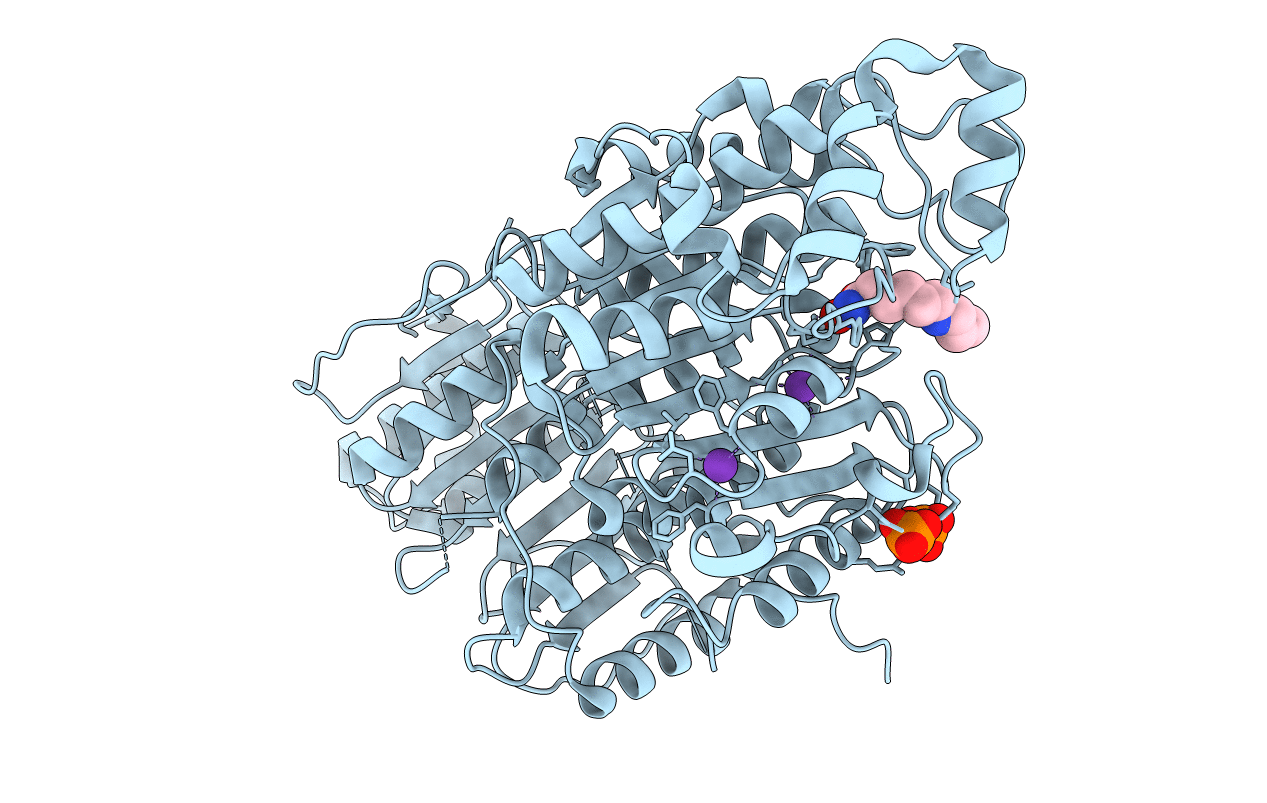
Title:
Crystal Structure of Danio rerio Histone Deacetylase 10 in Complex with 6-[(3-aminopropyl)amino]-N-hydroxyhexanamide
Biological Source:
Source Organism(s):
Danio rerio (Taxon ID: 7955)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.53 Å
R-Value Free:
0.24
R-Value Work:
0.20
R-Value Observed:
0.20
Space Group:
P 31 2 1


