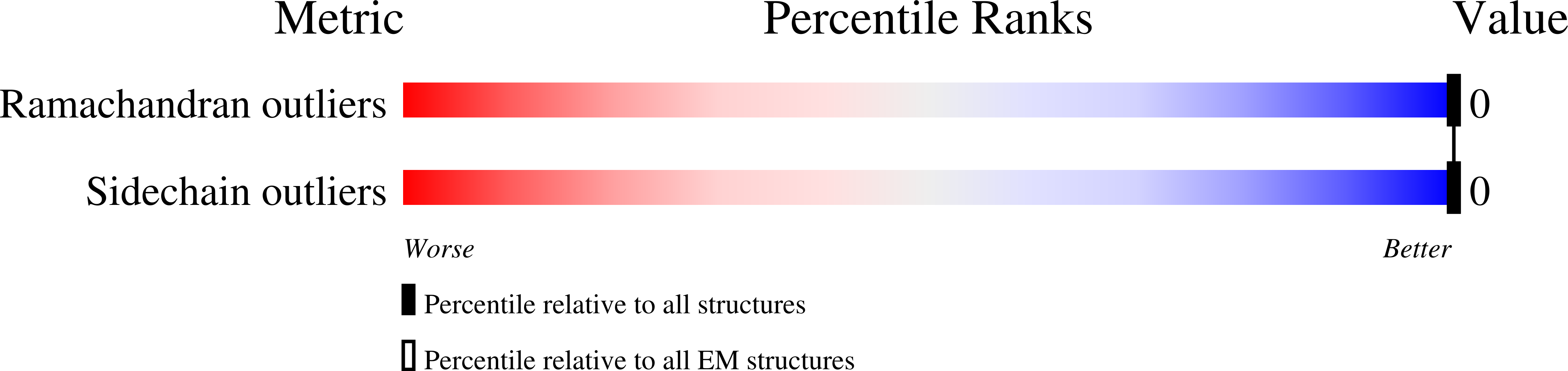
Deposition Date
2019-10-03
Release Date
2019-10-30
Last Version Date
2024-05-22
Entry Detail
PDB ID:
6T17
Keywords:
Title:
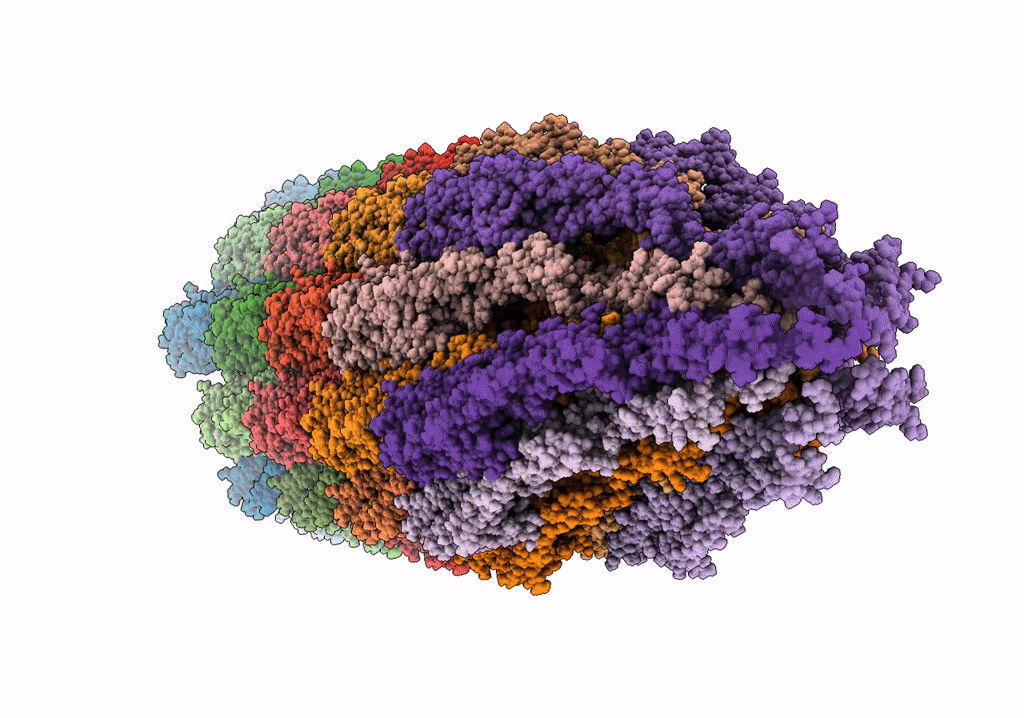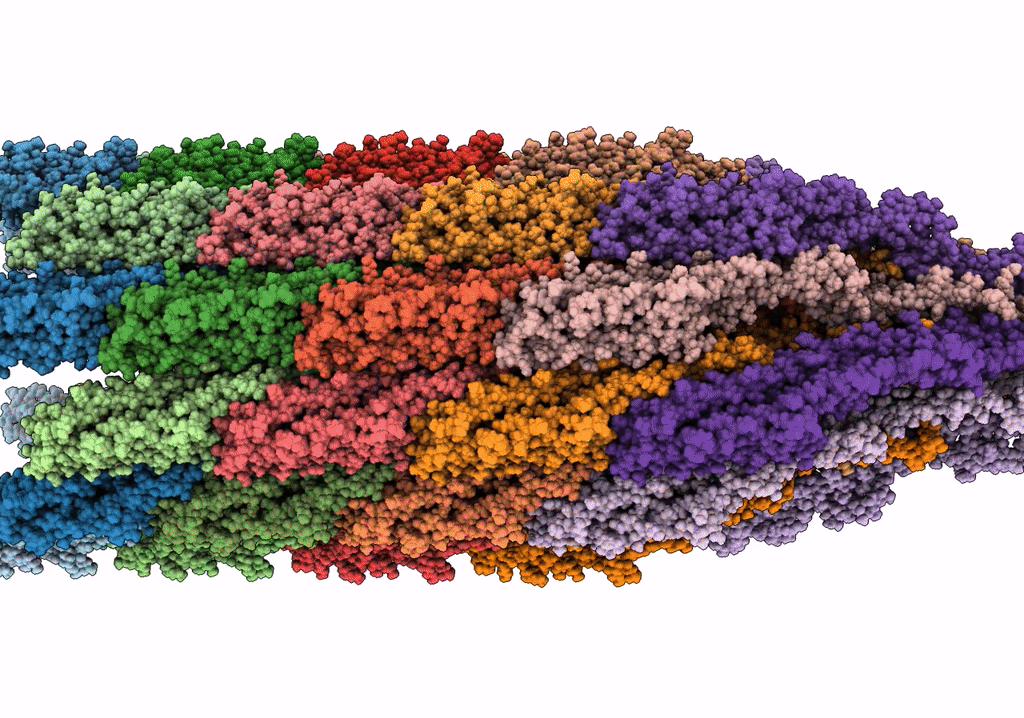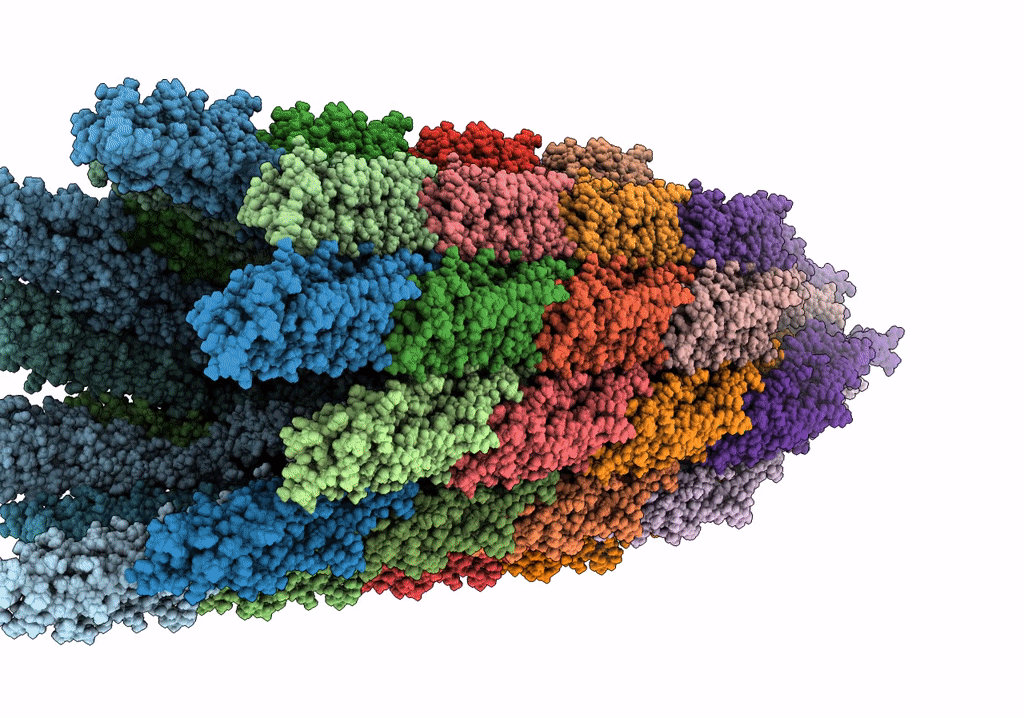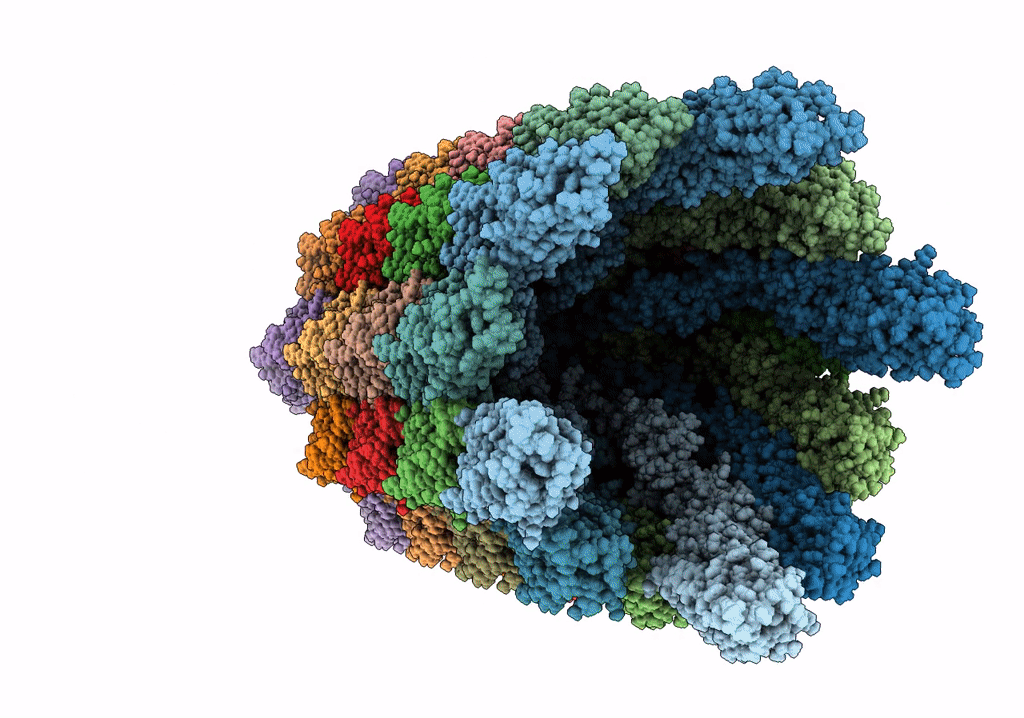
Cryo-EM structure of the wild-type flagellar filament of the Firmicute Kurthia
Biological Source:
Source Organism(s):
Kurthia sp. 11kri321 (Taxon ID: 1750719)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.80 Å
Aggregation State:
FILAMENT
Reconstruction Method:
HELICAL