Deposition Date
2019-07-05
Release Date
2019-07-17
Last Version Date
2024-05-15
Entry Detail
PDB ID:
6S7J
Keywords:
Title:
Native crystal structure of ergothioneine degrading enzyme Ergothionase from Treponema denticola
Biological Source:
Source Organism(s):
Treponema denticola SP33 (Taxon ID: 999437)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.20 Å
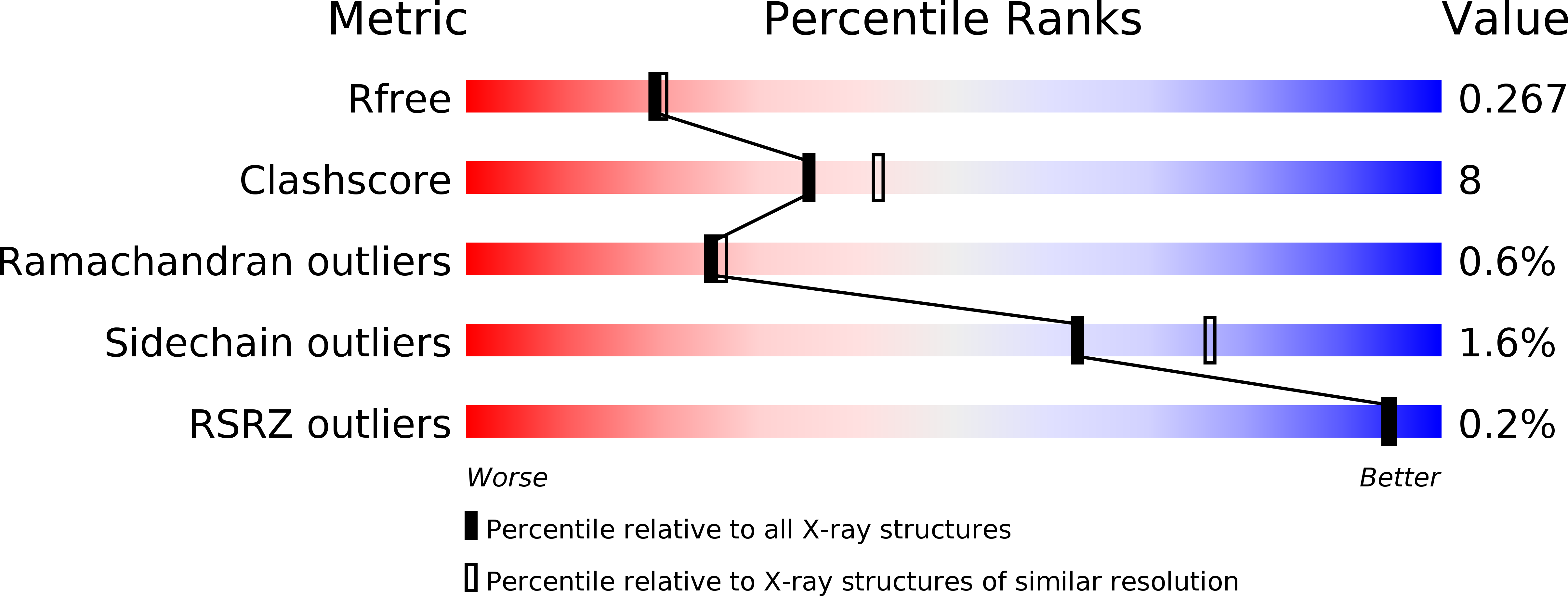
R-Value Free:
0.26
R-Value Work:
0.21
R-Value Observed:
0.21
Space Group:
P 1