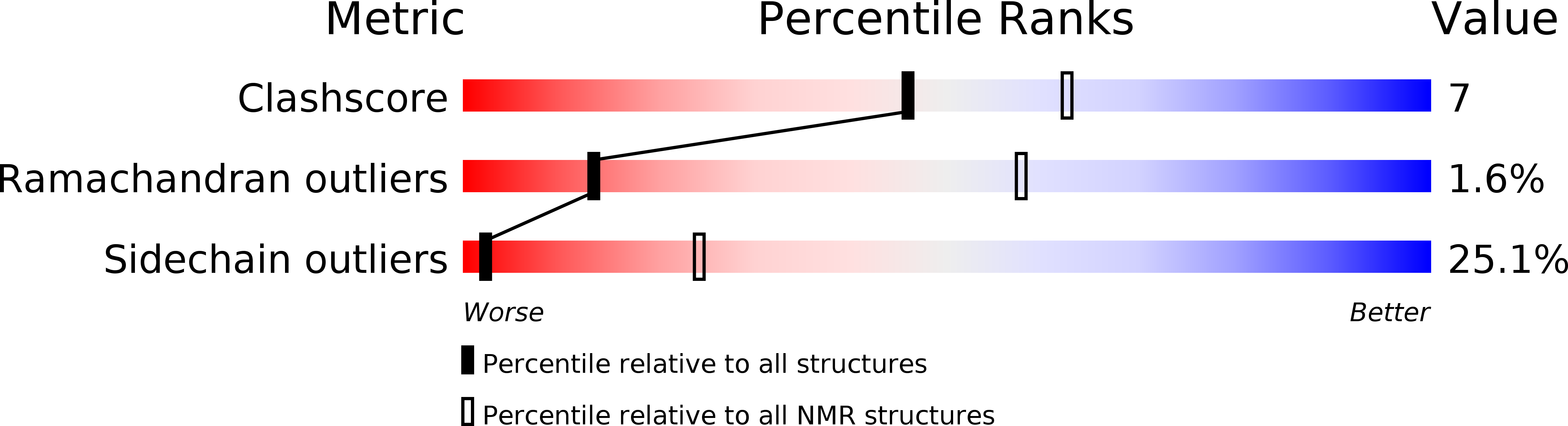Deposition Date
2019-04-26
Release Date
2019-06-05
Last Version Date
2024-05-01
Entry Detail
PDB ID:
6OQH
Keywords:
Title:
Solution NMR structure of a quiet outer membrane protein G Nanopore (OmpG mutant: Delta-L6-D215)
Biological Source:
Source Organism:
Escherichia coli (strain K12) (Taxon ID: 83333)
Host Organism:
Method Details:
Experimental Method:
Conformers Calculated:
200
Conformers Submitted:
10
Selection Criteria:
structures with the lowest energy