Deposition Date
2020-01-22
Release Date
2021-03-10
Last Version Date
2023-11-29
Entry Detail
PDB ID:
6LTC
Keywords:
Title:
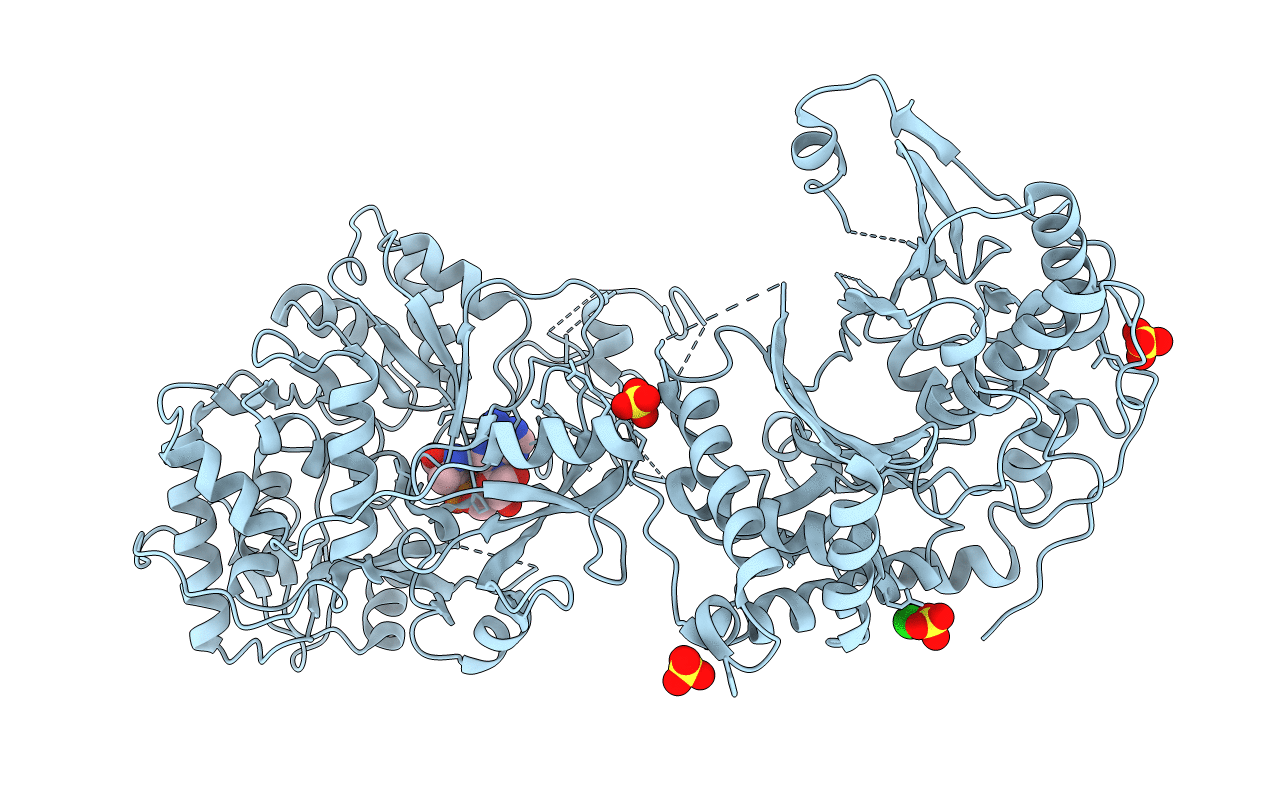
Crystal Structure of Nonribosomal peptide synthetases (NRPS), FmoA3 (S1046A)-alpha-methyl-L-serine-AMP bound form
Biological Source:
Source Organism(s):
Streptomyces sp. Sp080513GE-23 (Taxon ID: 630397)
Expression System(s):
Method Details:
Experimental Method:
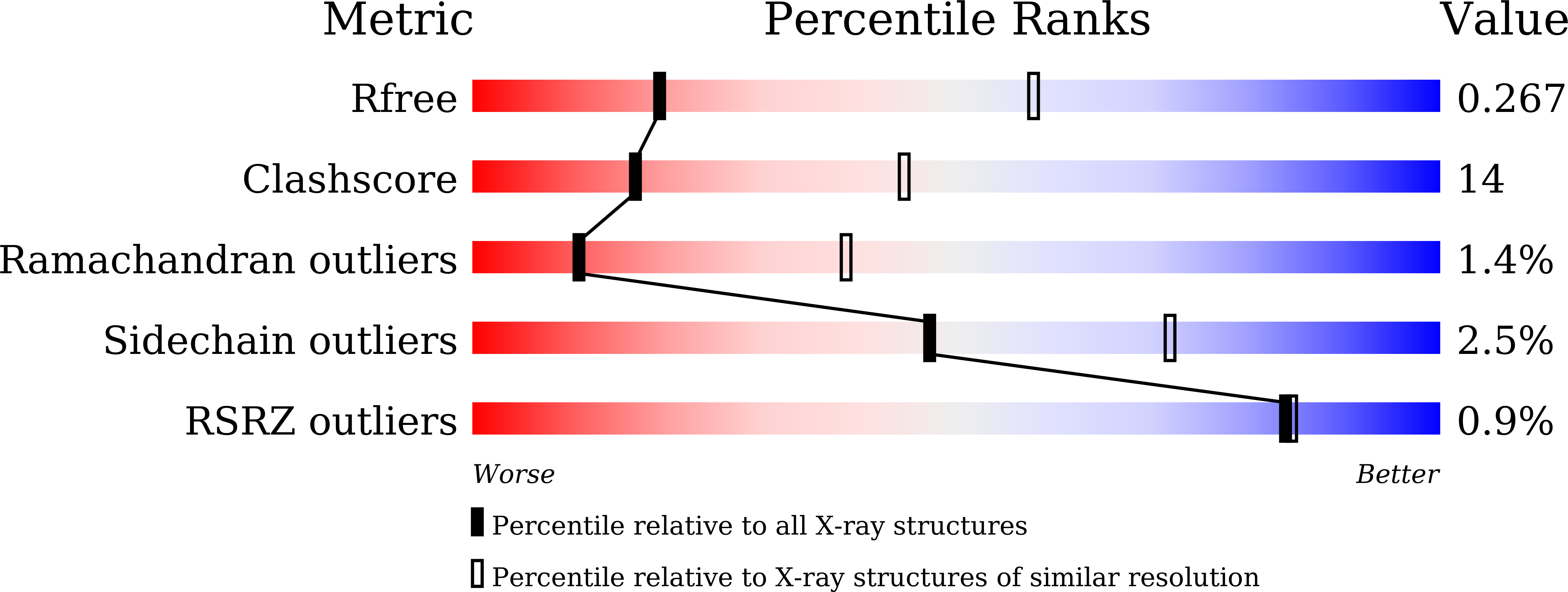
Resolution:
3.30 Å
R-Value Free:
0.26
R-Value Work:
0.21
R-Value Observed:
0.22
Space Group:
C 1 2 1