Deposition Date
2019-05-28
Release Date
2019-08-28
Last Version Date
2024-10-30
Entry Detail
PDB ID:
6K5A
Keywords:
Title:
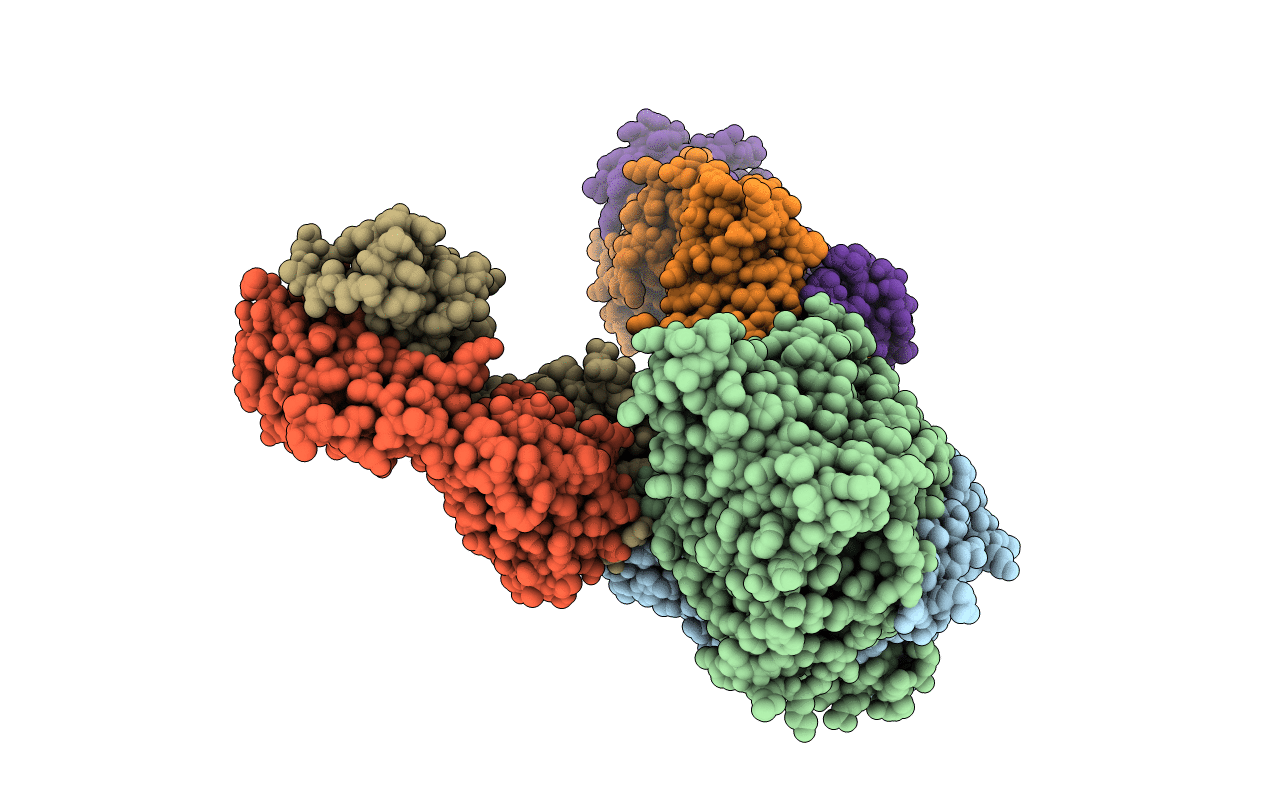
Crystal structure of the E148D/R147A/F317A mutant in presence of 200 mM NaBr
Biological Source:
Source Organism(s):
Escherichia coli MS 117-3 (Taxon ID: 749539)
Mus musculus (Taxon ID: 10090)
Mus musculus (Taxon ID: 10090)
Expression System(s):
Method Details:
Experimental Method:
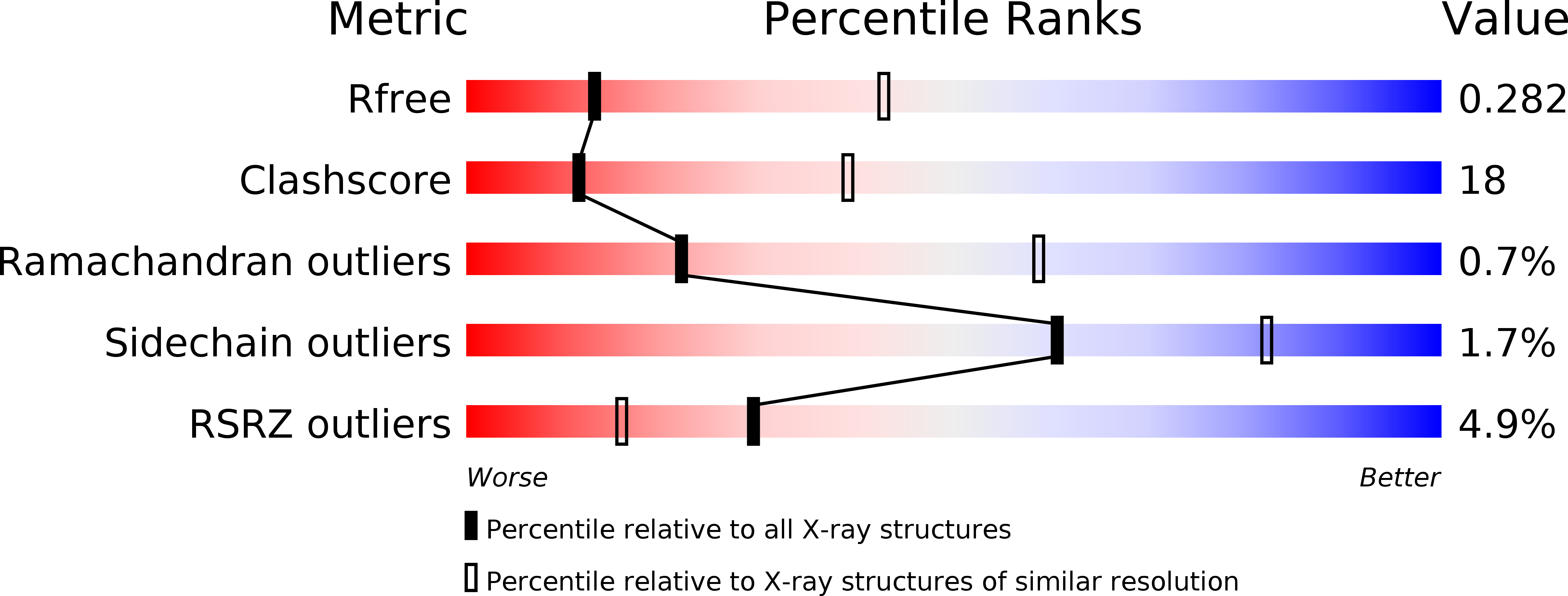
Resolution:
3.16 Å
R-Value Free:
0.27
R-Value Work:
0.22
R-Value Observed:
0.22
Space Group:
C 1 2 1