Deposition Date
2018-07-10
Release Date
2019-05-15
Last Version Date
2024-11-20
Entry Detail
PDB ID:
6H13
Keywords:
Title:
Crystal structure of TcACHE complexed to1-(4-((Methyl((1-(2-((1,2,3,4-tetrahydroacridin-9-yl)amino)ethyl)-1H-1,2,3-triazol-4-yl)methyl)amino)methyl)pyridin-2-yl)-3-(6-oxo-1,2,3,4,6,10b-hexahydropyrido[2,1-a]isoindol-10-yl)urea
Biological Source:
Source Organism(s):
Tetronarce californica (Taxon ID: 7787)
Method Details:
Experimental Method:
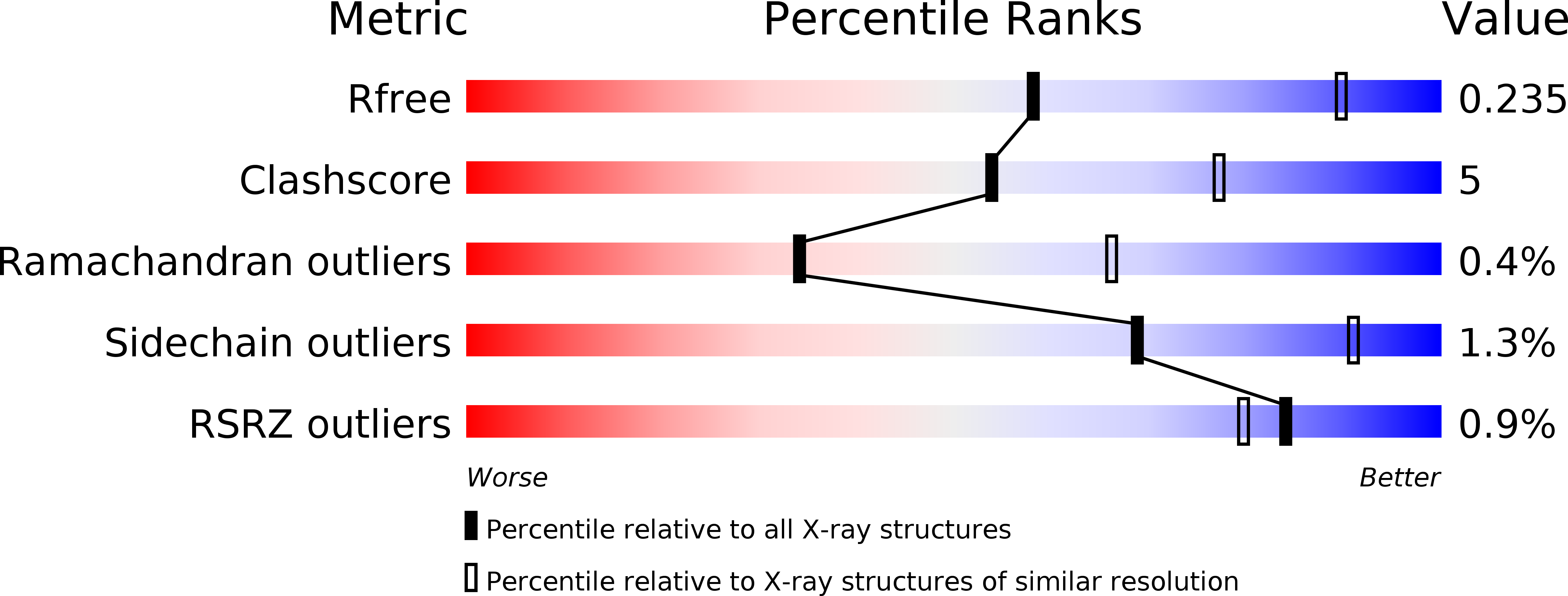
Resolution:
2.80 Å
R-Value Free:
0.23
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 21 21 21