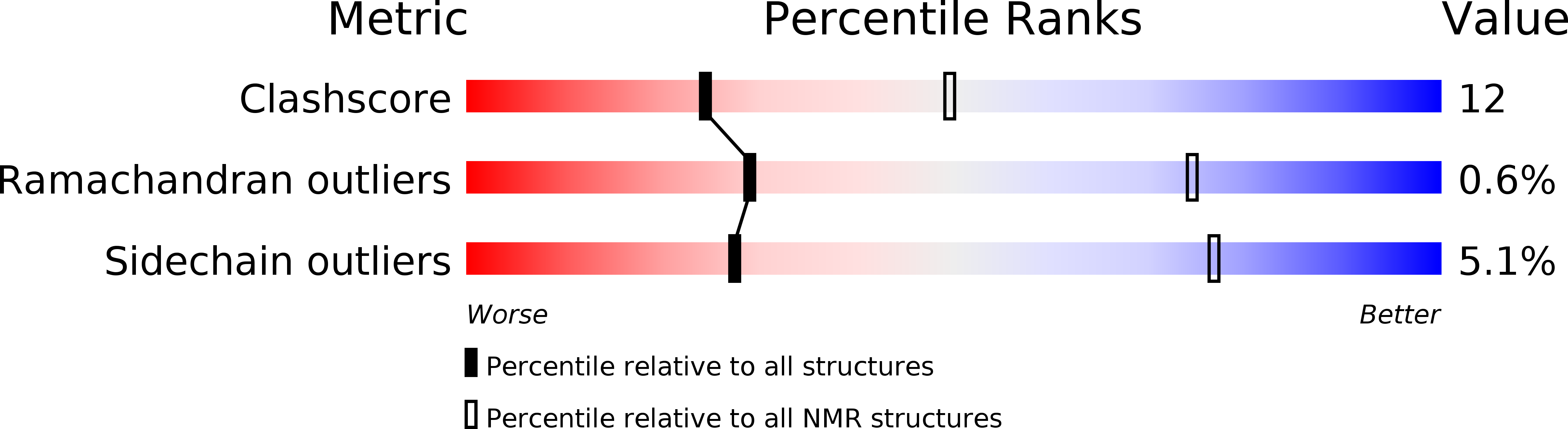
Deposition Date
2017-06-01
Release Date
2018-06-06
Last Version Date
2024-10-23
Entry Detail
PDB ID:
5W0Y
Keywords:
Title:
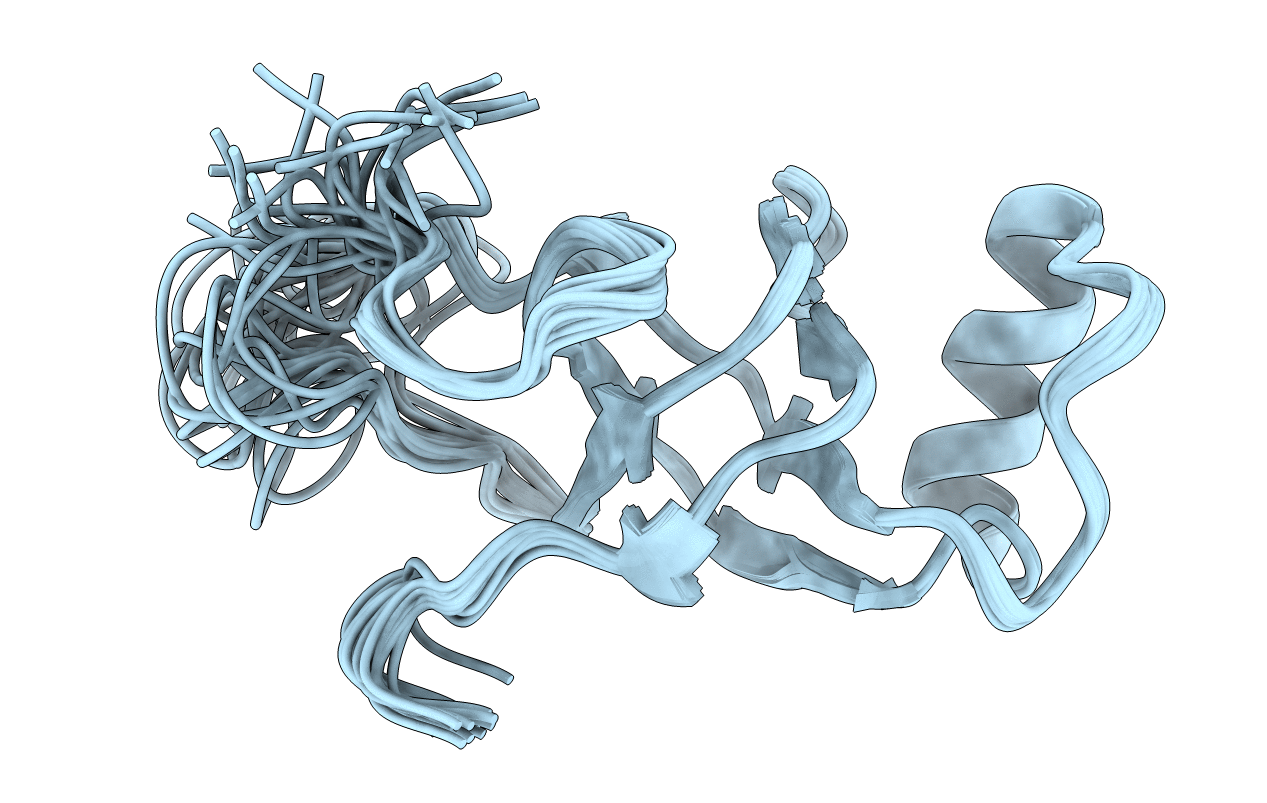
Solution NMR Structure of a Class I Hydrophobin from Serpula lacrymans
Biological Source:
Source Organism(s):
Serpula lacrymans var. lacrymans (Taxon ID: 578457)
Expression System(s):
Method Details:
Experimental Method:
Conformers Calculated:
100
Conformers Submitted:
20
Selection Criteria:
structures with the lowest energy