Deposition Date
2016-11-12
Release Date
2017-06-28
Last Version Date
2023-10-04
Entry Detail
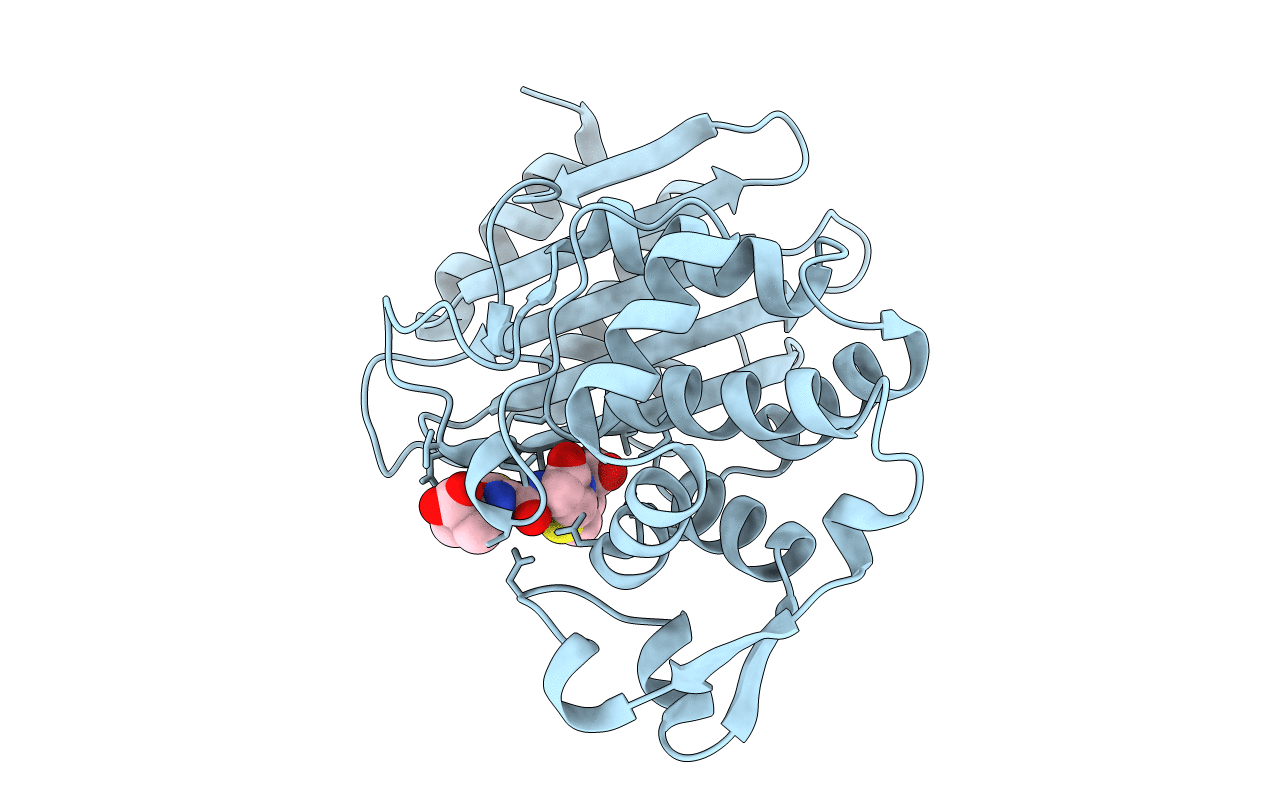
PDB ID:
5TWE
Keywords:
Title:
CTX-M-14 P167S:S70G mutant enzyme crystallized with ceftazidime
Biological Source:
Source Organism:
Escherichia coli (Taxon ID: 562)
Host Organism:
Method Details:
Experimental Method:
Resolution:
1.50 Å
R-Value Free:
0.16
R-Value Work:
0.15
R-Value Observed:
0.15
Space Group:
P 32 2 1


