Deposition Date
2017-03-06
Release Date
2017-05-24
Last Version Date
2025-10-01
Entry Detail
PDB ID:
5NCO
Keywords:
Title:
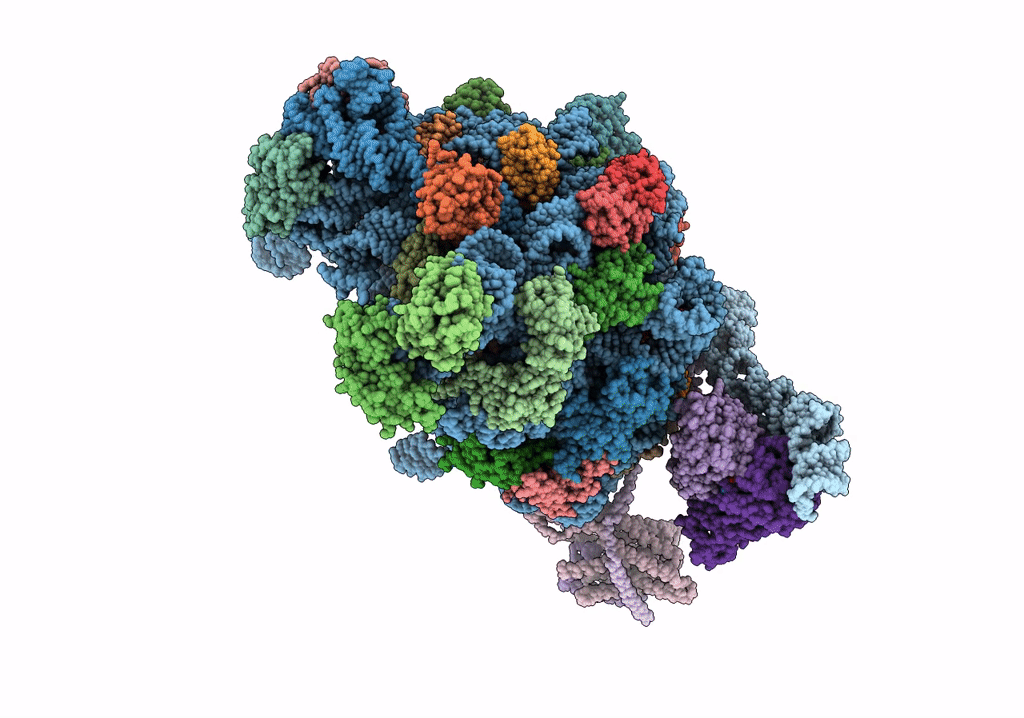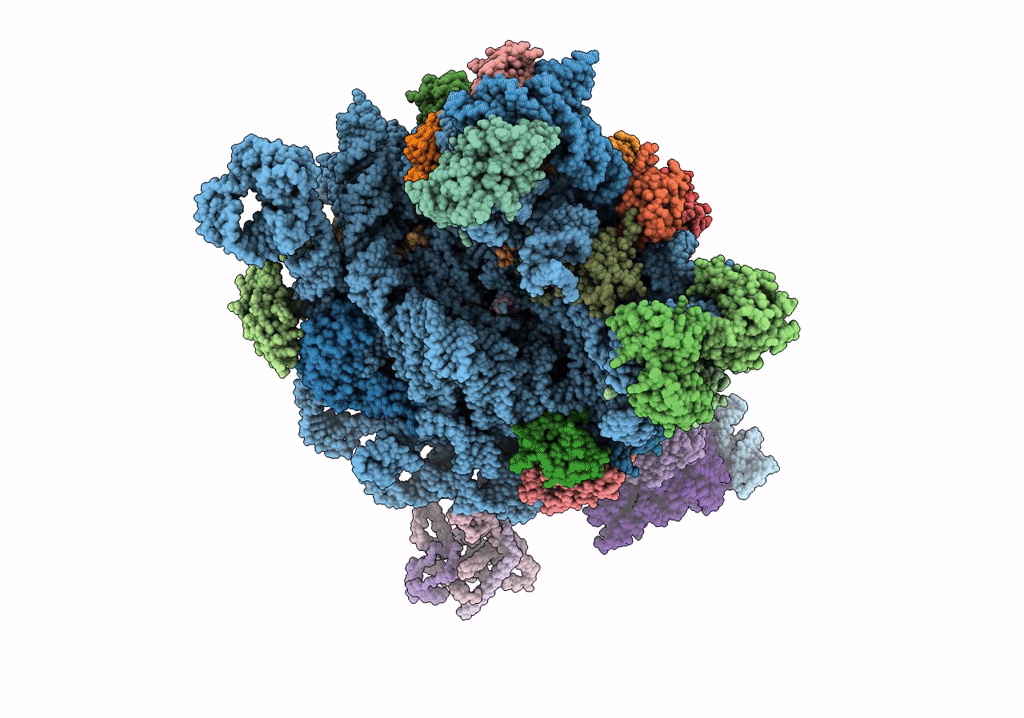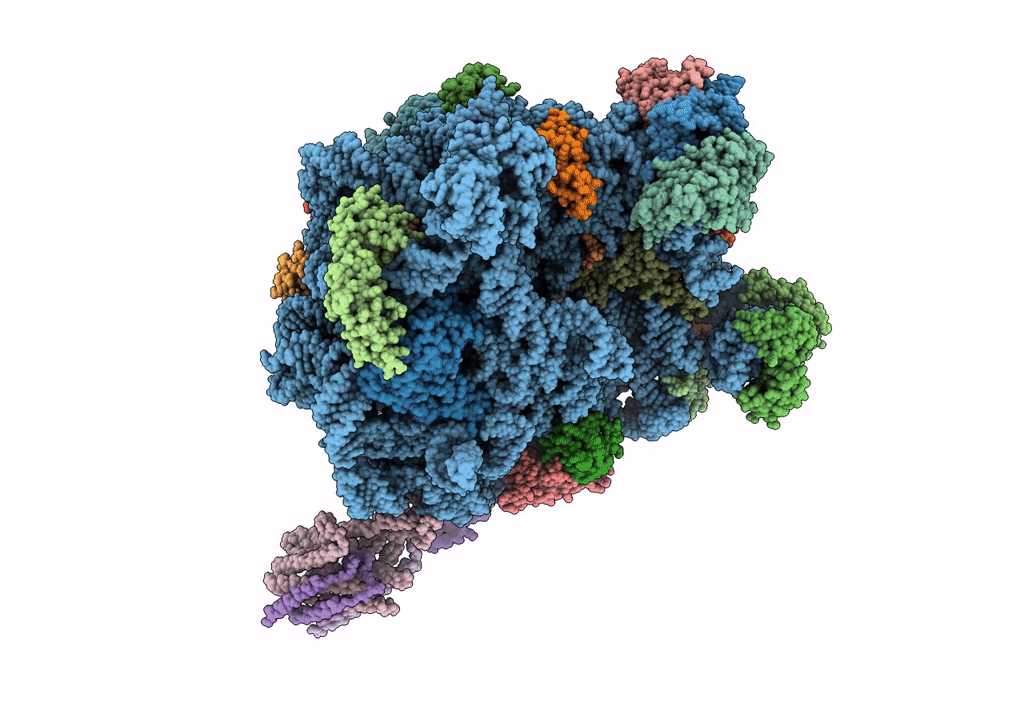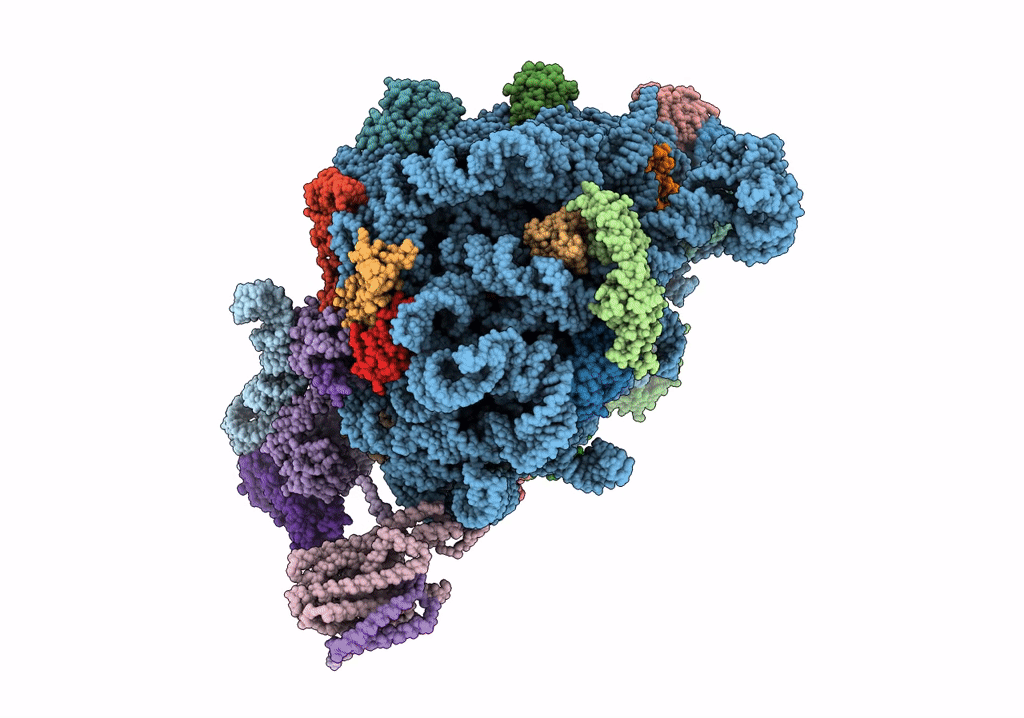
Quaternary complex between SRP, SR, and SecYEG bound to the translating ribosome
Biological Source:
Source Organism:
Escherichia coli (Taxon ID: 562)
Host Organism:
Method Details:
Experimental Method:
Resolution:
4.80 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE


