Deposition Date
2015-07-15
Release Date
2015-09-09
Last Version Date
2023-09-27
Entry Detail
PDB ID:
5CJW
Keywords:
Title:
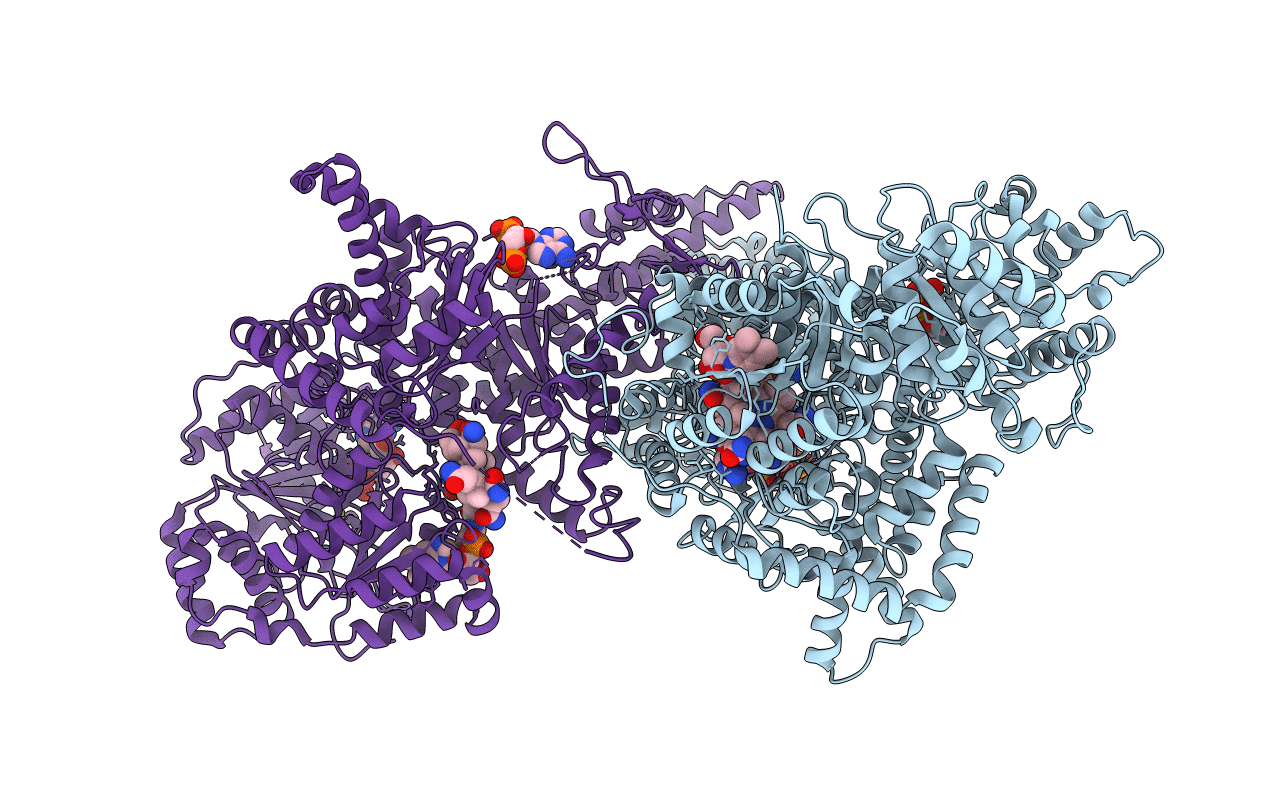
Isobutyryl-CoA mutase fused with bound adenosylcobalamin, GDP, Mg (holo-IcmF/GDP), and substrate pivalyl-coenzyme A
Biological Source:
Source Organism(s):
Expression System(s):
Method Details:
Experimental Method:
Resolution:
3.40 Å
R-Value Free:
0.20
R-Value Work:
0.18
R-Value Observed:
0.18
Space Group:
H 3 2


