Deposition Date
2015-06-04
Release Date
2015-08-05
Last Version Date
2024-10-23
Entry Detail
PDB ID:
5BV0
Keywords:
Title:
Crystal Structure of a Complex Between the SNARE Nyv1 and the HOPS Vps33-Vps16 subcomplex from Chaetomium thermophilum
Biological Source:
Source Organism(s):
Chaetomium thermophilum (strain DSM 1495 / CBS 144.50 / IMI 039719) (Taxon ID: 759272)
Chaetomium thermophilum (Taxon ID: 209285)
Chaetomium thermophilum (Taxon ID: 209285)
Expression System(s):
Method Details:
Experimental Method:
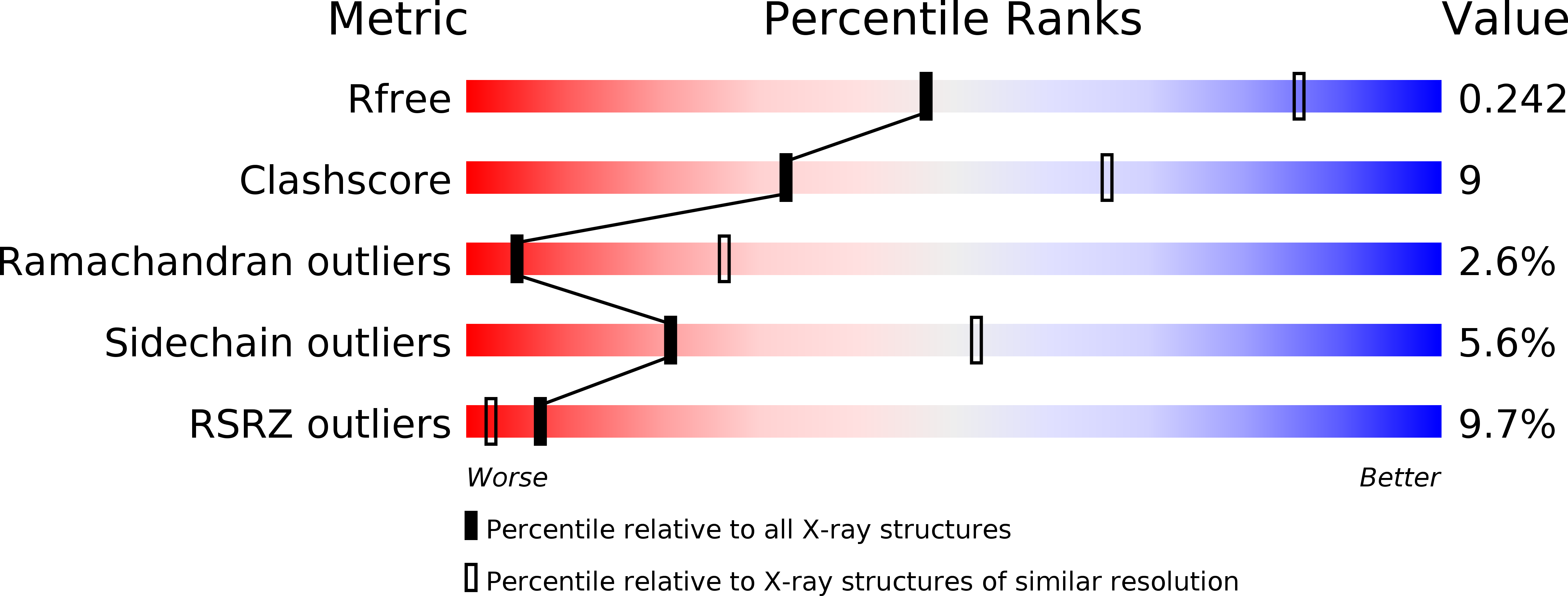
Resolution:
3.10 Å
R-Value Free:
0.24
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 21 21 2