Deposition Date
2014-06-23
Release Date
2015-07-08
Last Version Date
2023-09-27
Entry Detail
PDB ID:
4TU5
Keywords:
Title:
Staphylococcus aureus Dihydrofolate Reductase complexed with NADPH and 6-ETHYL-5-[(3S)-3-[3-METHOXY-5-(PYRIDIN-4-YL)PHENYL]BUT-1-YN-1-YL]PYRIMIDINE-2,4-DIAMINE (UCP1062)
Biological Source:
Source Organism(s):
Staphylococcus aureus (Taxon ID: 273036)
Expression System(s):
Method Details:
Experimental Method:
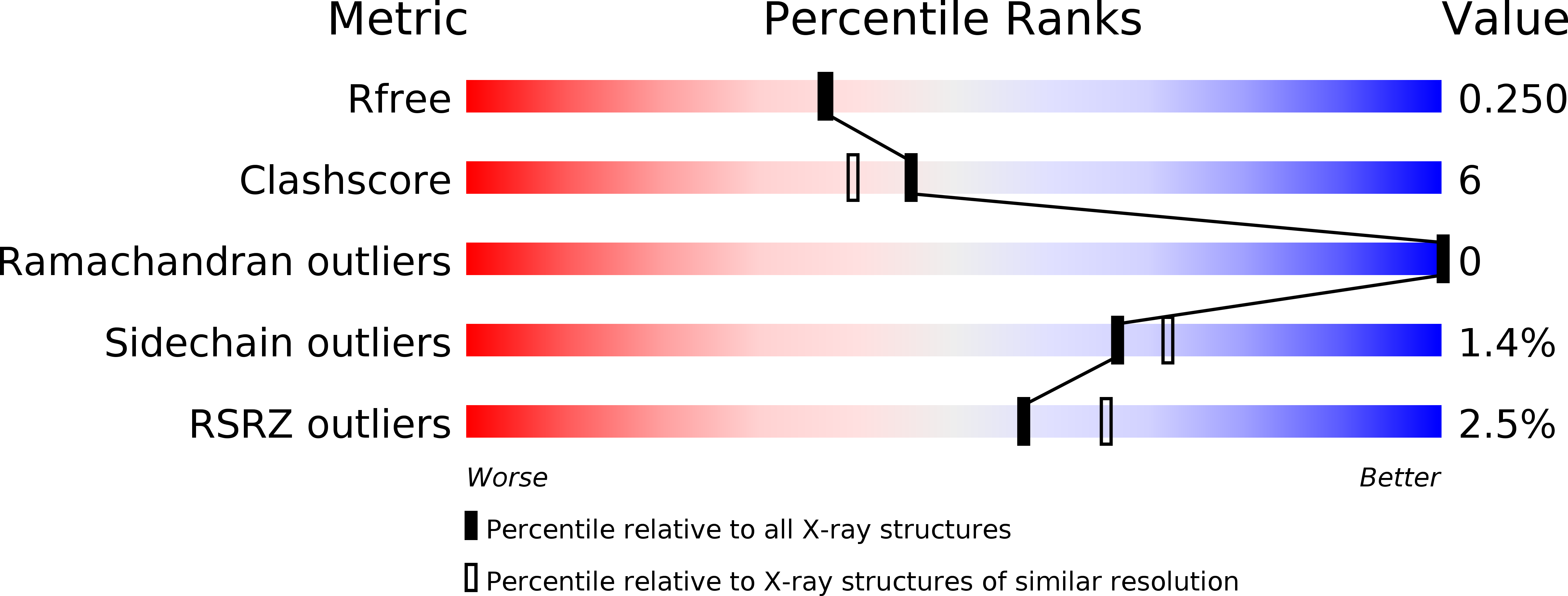
Resolution:
2.16 Å
R-Value Free:
0.24
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 61 2 2