Deposition Date
2014-04-23
Release Date
2014-07-23
Last Version Date
2024-10-16
Entry Detail
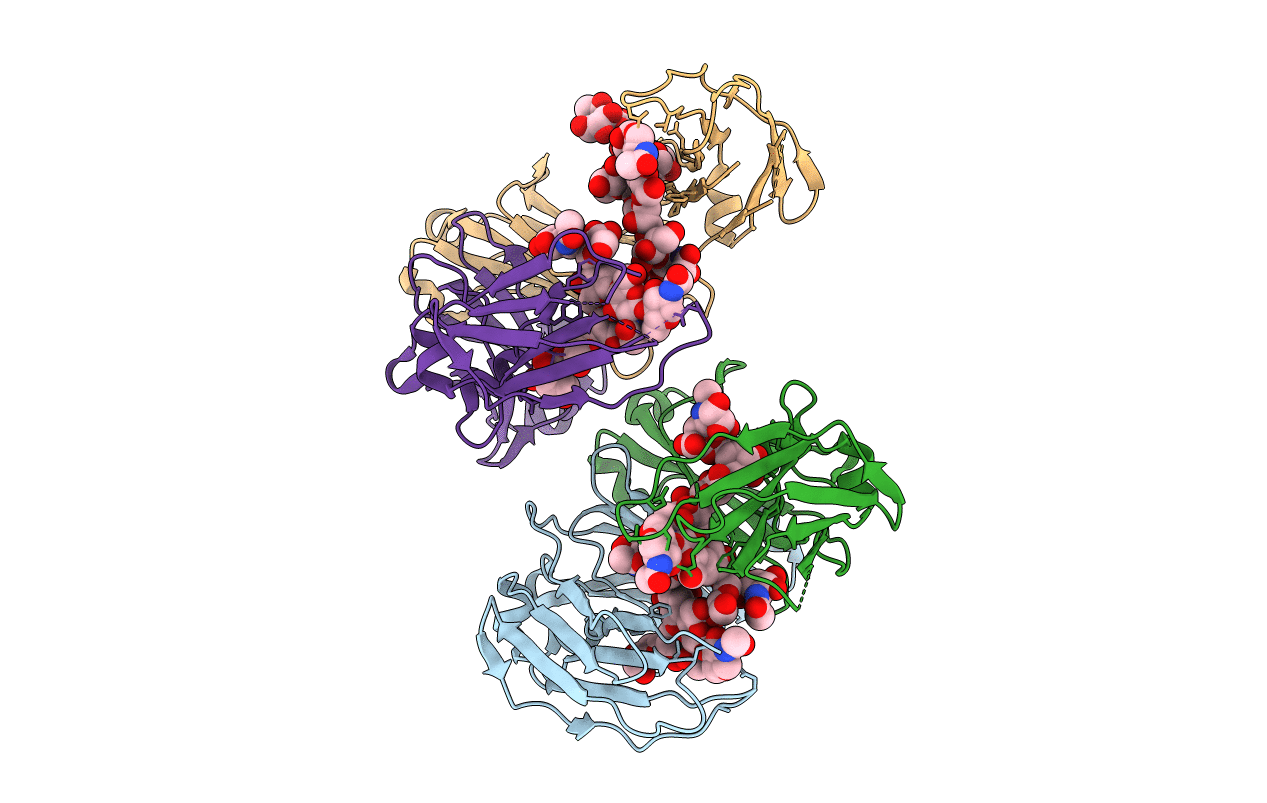
PDB ID:
4Q6Y
Keywords:
Title:
Crystal structure of a chemoenzymatic glycoengineered disialylated Fc (di-sFc)
Biological Source:
Source Organism:
Homo sapiens (Taxon ID: 9606)
Host Organism:
Method Details:
Experimental Method:
Resolution:
3.00 Å
R-Value Free:
0.28
R-Value Work:
0.26
Space Group:
P 1 21 1


