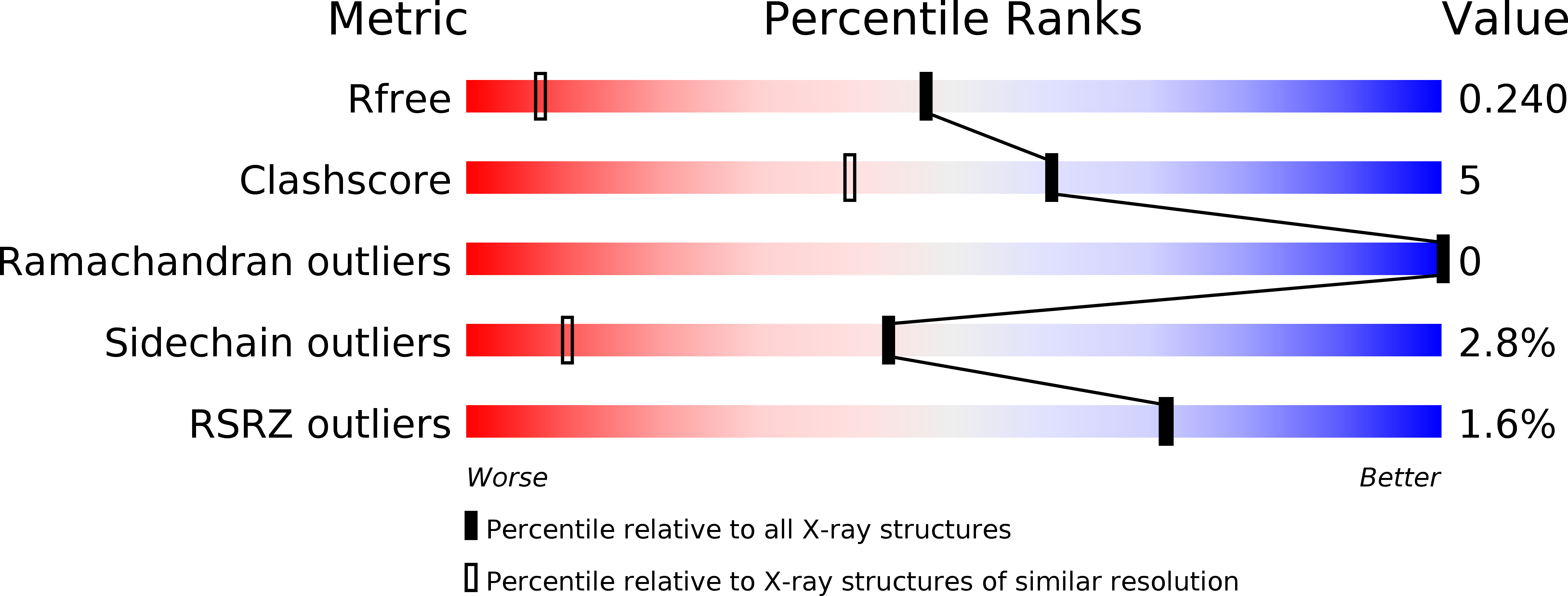
Abstact
Nitronate monooxygenase (NMO) oxidizes the mitochondrial toxin propionate 3-nitronate (P3N) to malonate semialdehyde. The enzyme has been previously characterized biochemically in fungi, but no structural information is available. Based on amino acid similarity 4,985 genes are annotated in the GenBank(TM) as NMO. Of these, 4,424 (i.e. 89%) are bacterial genes, including several Pseudomonads that have been shown to use P3N as growth substrate. Here, we have cloned and expressed the gene pa4202 of Pseudomonas aeruginosa PAO1, purified the resulting protein, and characterized it. The enzyme is active on P3N and other alkyl nitronates, but cannot oxidize nitroalkanes. P3N is the best substrate at pH 7.5 and atmospheric oxygen with k(cat)(app)/K(m)(app) of 12 × 10(6) M(-1) s(-1), k(cat)(app) of 1300 s(-1), and K(m)(app) of 110 μm. Anerobic reduction of the enzyme with P3N yields a flavosemiquinone, which is formed within 7.5 ms, consistent with this species being a catalytic intermediate. Absorption spectroscopy, mass spectrometry, and x-ray crystallography demonstrate a tightly, non-covalently bound FMN in the active site of the enzyme. Thus, PA4202 is the first NMO identified and characterized in bacteria. The x-ray crystal structure of the enzyme was solved at 1.44 Å, showing a TIM barrel-fold. Four motifs in common with the biochemically characterized NMO from Cyberlindnera saturnus are identified in the structure of bacterial NMO, defining Class I NMO, which includes bacterial, fungal, and two animal NMOs. Notably, the only other NMO from Neurospora crassa for which biochemical evidence is available lacks the four motifs, defining Class II NMO.