Deposition Date
2012-09-06
Release Date
2013-02-13
Last Version Date
2023-09-13
Entry Detail
PDB ID:
4GZZ
Keywords:
Title:
Crystal structures of bacterial RNA Polymerase paused elongation complexes
Biological Source:
Source Organism(s):
Thermus thermophilus (Taxon ID: 300852)
synthetic (Taxon ID: 32630)
synthetic (Taxon ID: 32630)
Expression System(s):
Method Details:
Experimental Method:
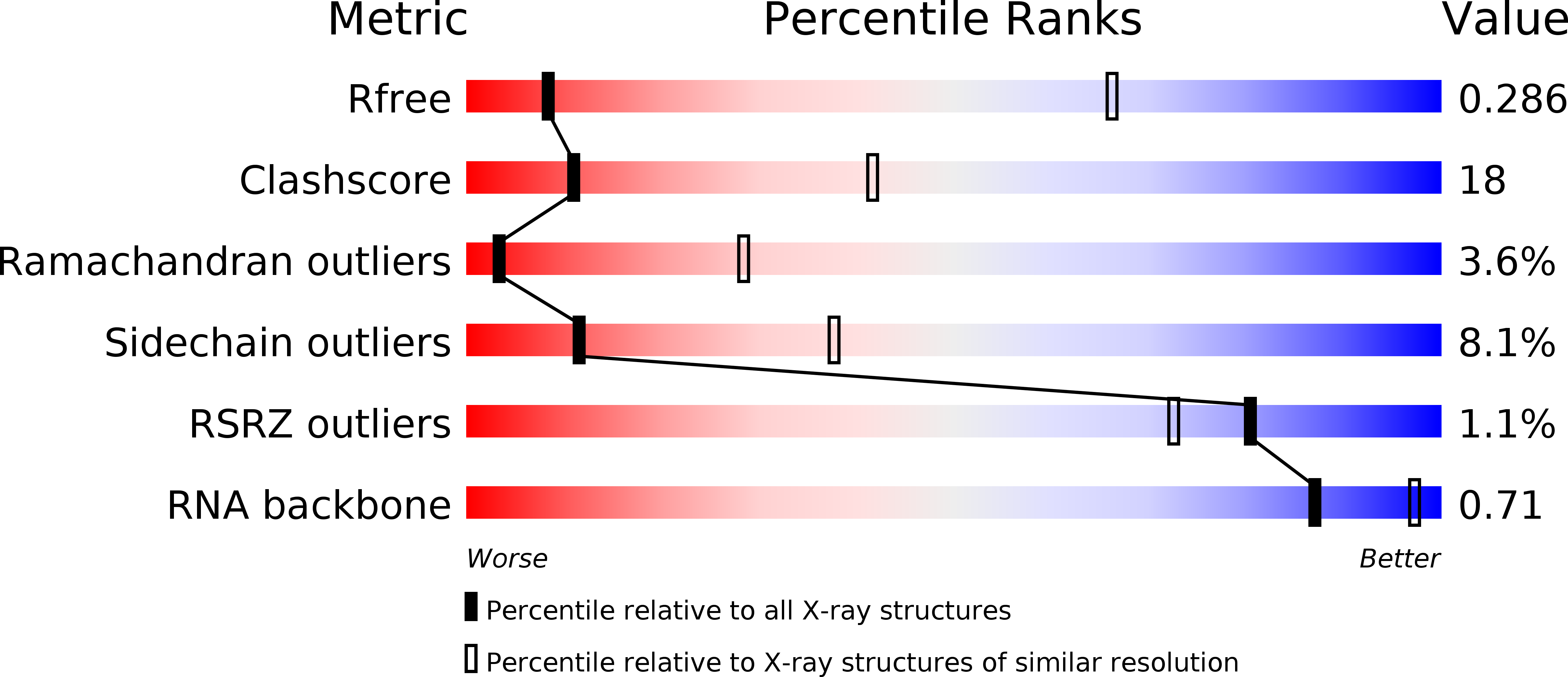
Resolution:
4.29 Å
R-Value Free:
0.28
R-Value Work:
0.23
R-Value Observed:
0.23
Space Group:
H 3