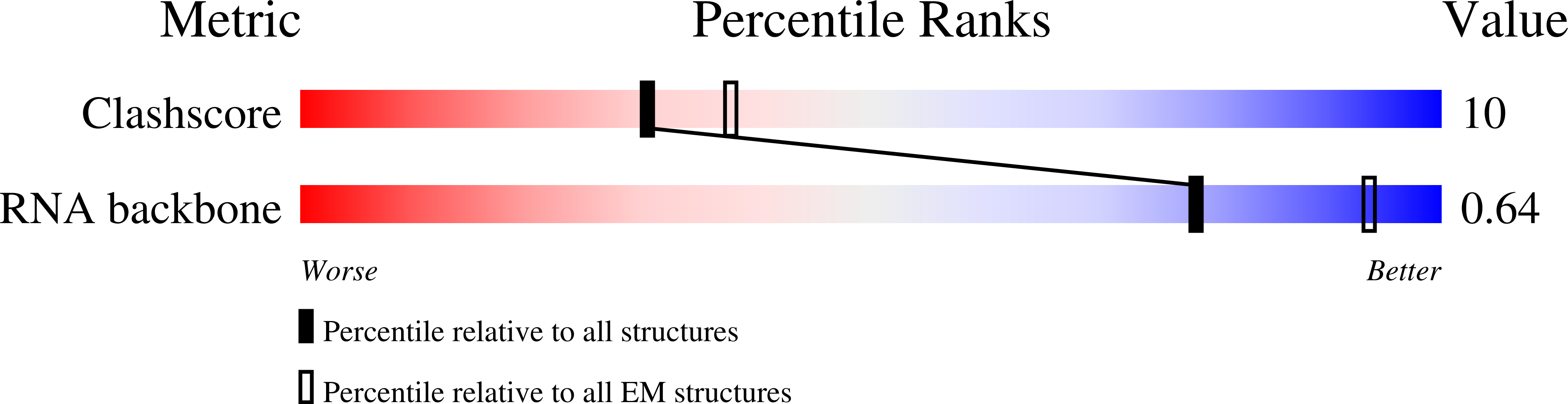
Deposition Date
2013-09-07
Release Date
2013-10-30
Last Version Date
2024-05-08
Entry Detail
PDB ID:
4C4Q
Keywords:
Title:
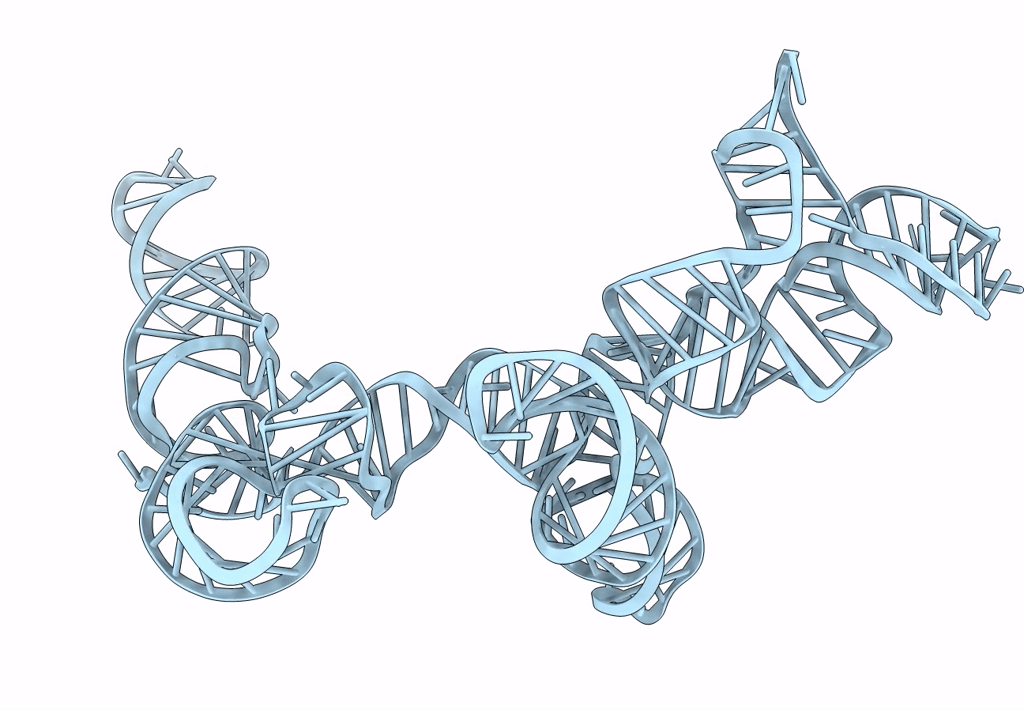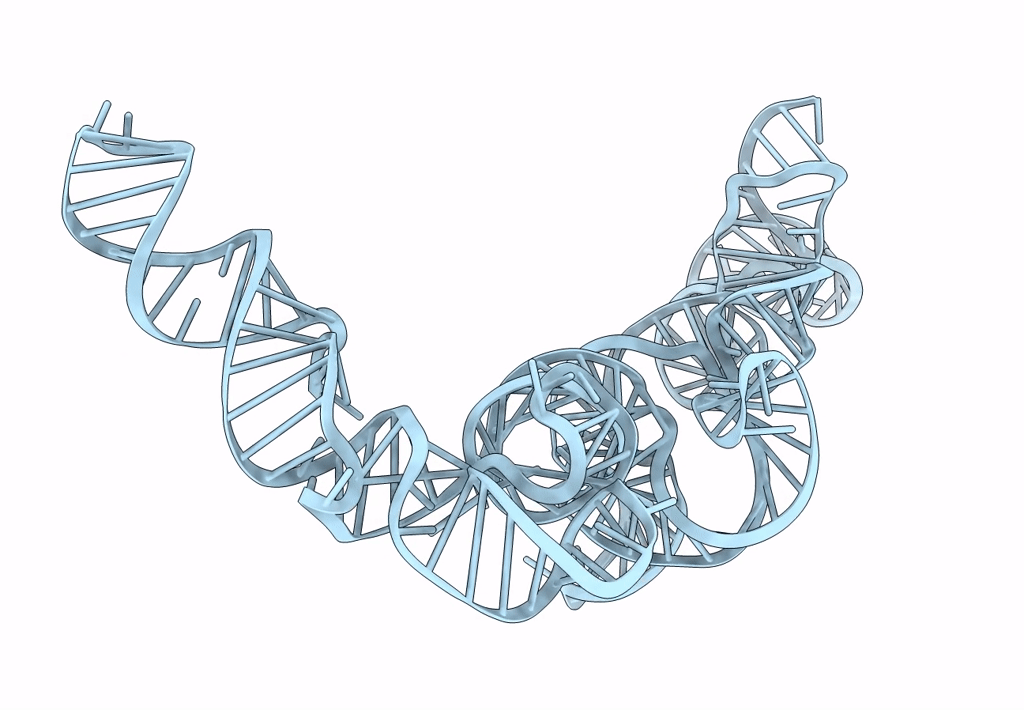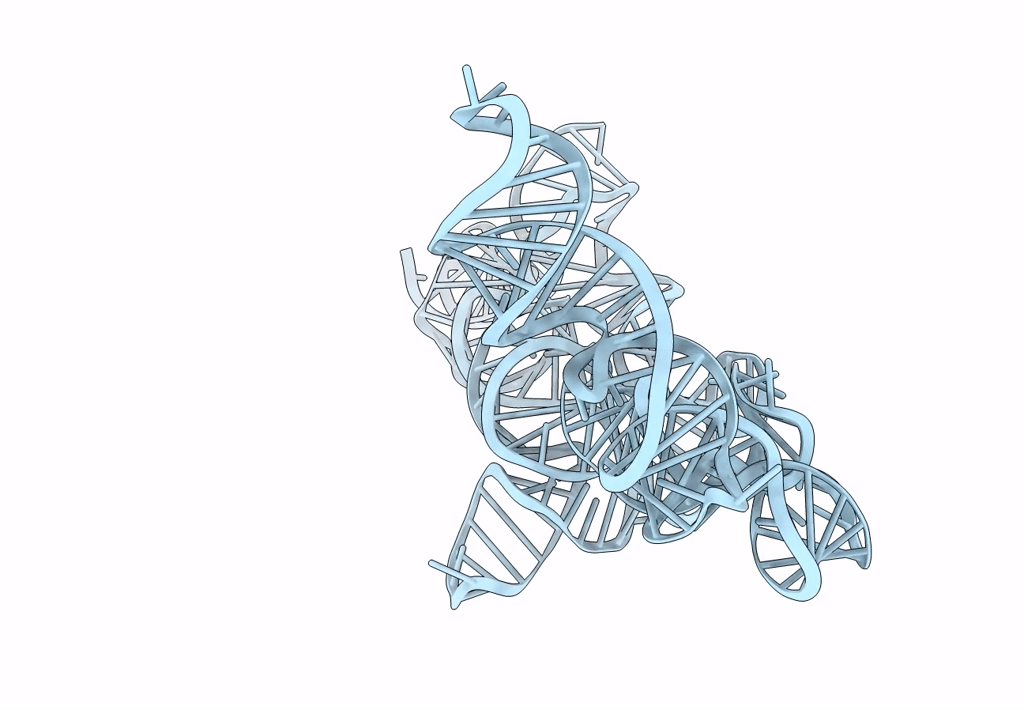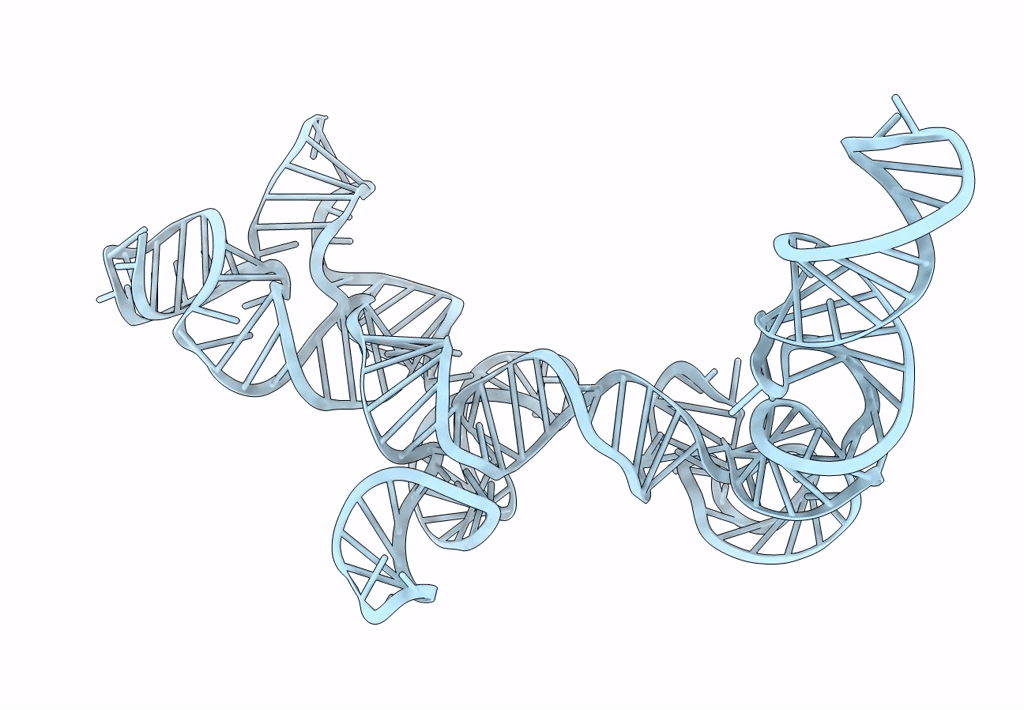
Cryo-EM map of the CSFV IRES in complex with the small ribosomal 40S subunit and DHX29
Biological Source:
Source Organism(s):
CLASSICAL SWINE FEVER VIRUS (Taxon ID: 11096)
Method Details:
Experimental Method:
Resolution:
8.50 Å
Aggregation State:
PARTICLE
Reconstruction Method:
SINGLE PARTICLE