Deposition Date
2011-11-01
Release Date
2012-07-11
Last Version Date
2023-09-13
Entry Detail
PDB ID:
3UFV
Keywords:
Title:
Structure of rat nitric oxide synthase heme domain in complex with 4-methyl-6-(((3R,4R)-4-((5-(4-methylpyridin-2-yl)pentyl)oxy)pyrrolidin-3-yl)methyl)pyridin-2-amine
Biological Source:
Source Organism(s):
Rattus norvegicus (Taxon ID: 10116)
Expression System(s):
Method Details:
Experimental Method:
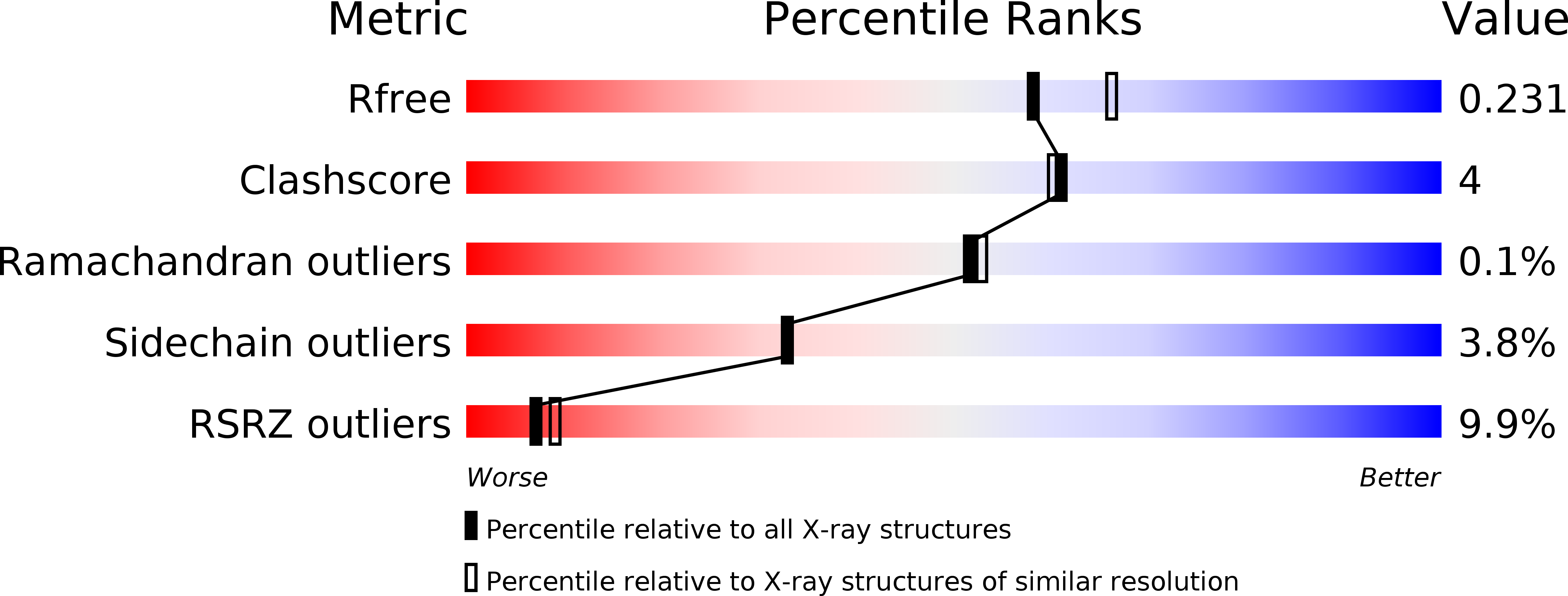
Resolution:
2.08 Å
R-Value Free:
0.21
R-Value Work:
0.17
R-Value Observed:
0.17
Space Group:
P 21 21 21