Deposition Date
2010-02-19
Release Date
2010-11-24
Last Version Date
2024-10-09
Entry Detail
PDB ID:
3LV3
Keywords:
Title:
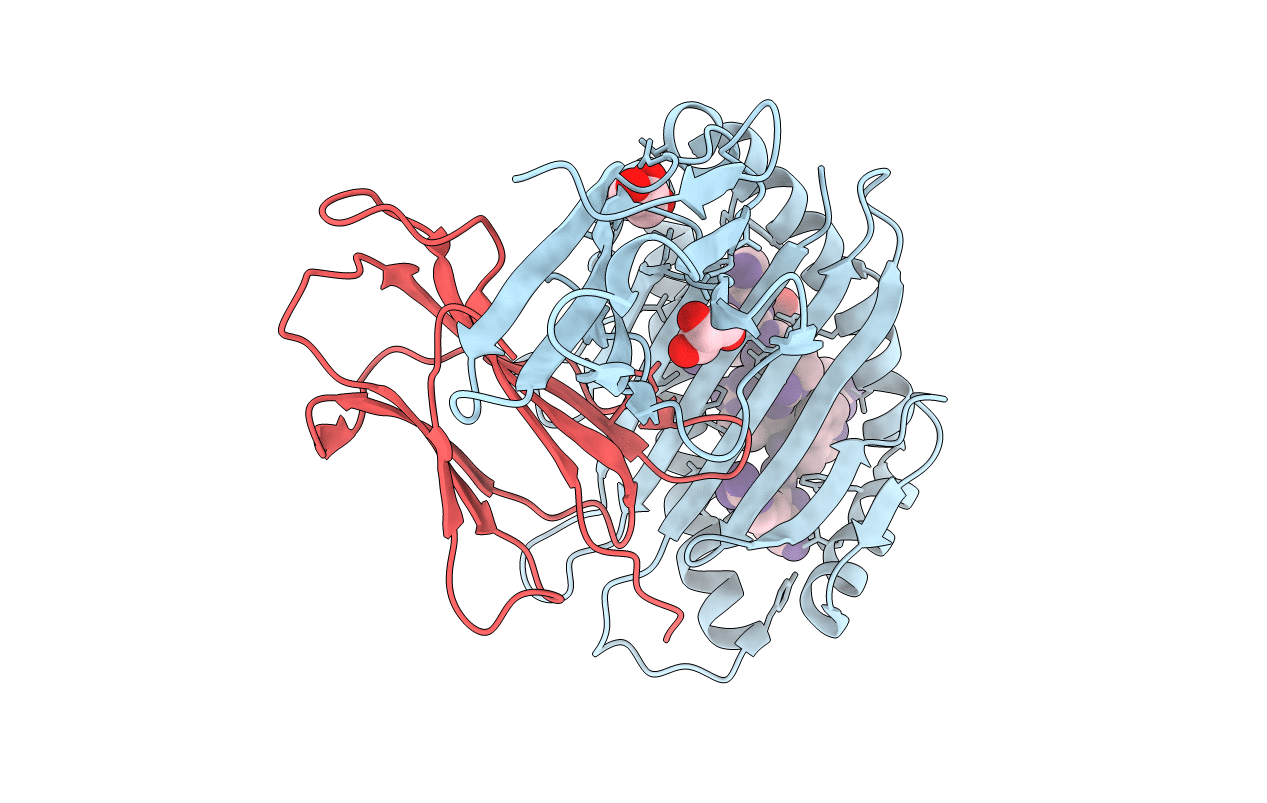
Crystal structure of HLA-B*2705 complexed with a peptide derived from the human voltage-dependent calcium channel alpha1 subunit (residues 513-521)
Biological Source:
Source Organism:
Homo sapiens (Taxon ID: 9606)
Host Organism:
Method Details:
Experimental Method:
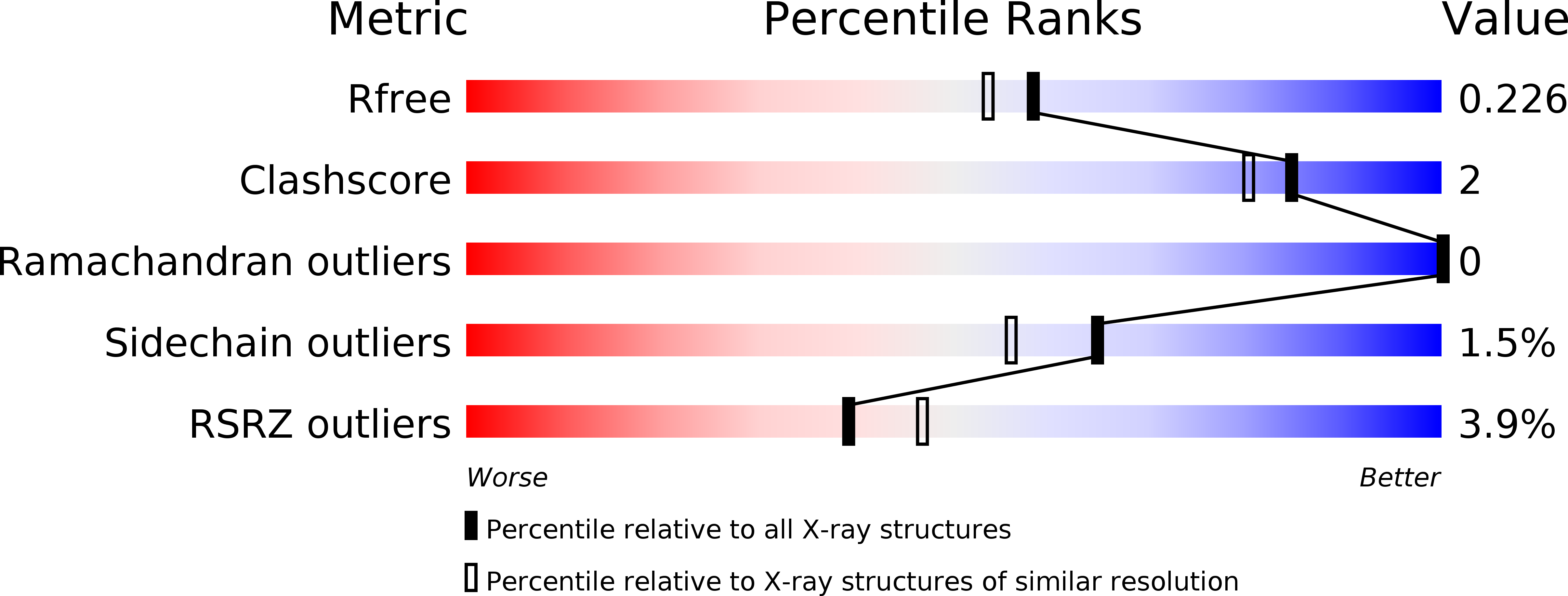
Resolution:
1.94 Å
R-Value Free:
0.22
R-Value Work:
0.17
R-Value Observed:
0.17
Space Group:
P 1 21 1