Deposition Date
2009-11-06
Release Date
2010-09-22
Last Version Date
2023-11-01
Entry Detail
PDB ID:
3KKR
Keywords:
Title:
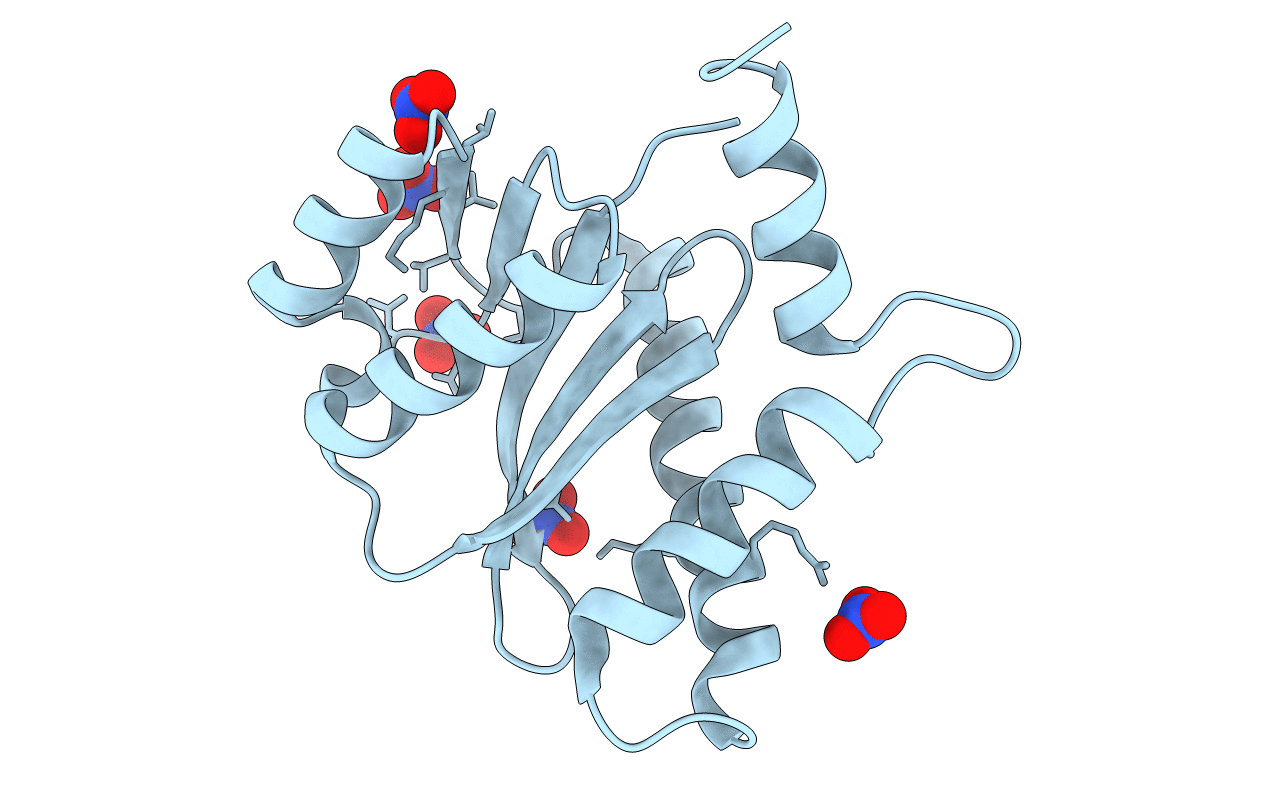
Crystal structure of catalytic core domain of BIV integrase in crystal form I
Biological Source:
Source Organism(s):
Bovine immunodeficiency virus (Taxon ID: 417296)
Expression System(s):
Method Details:
Experimental Method:
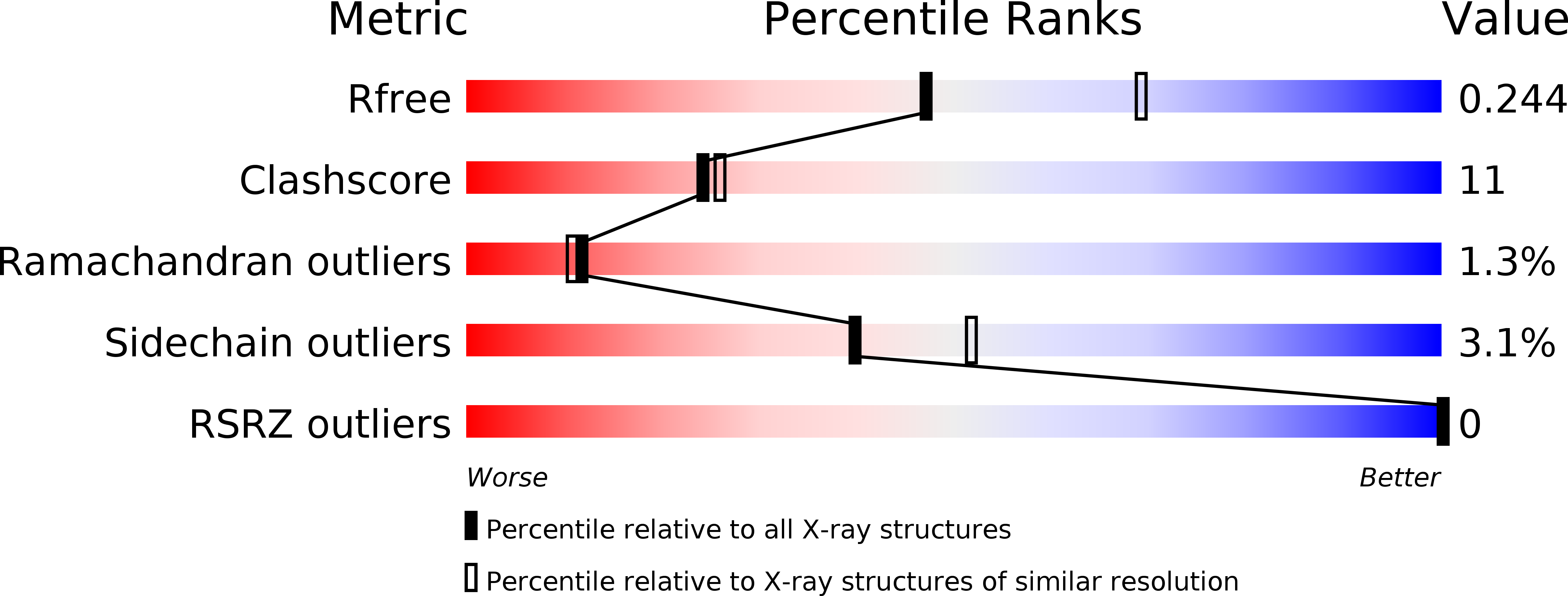
Resolution:
2.45 Å
R-Value Free:
0.24
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 41 21 2