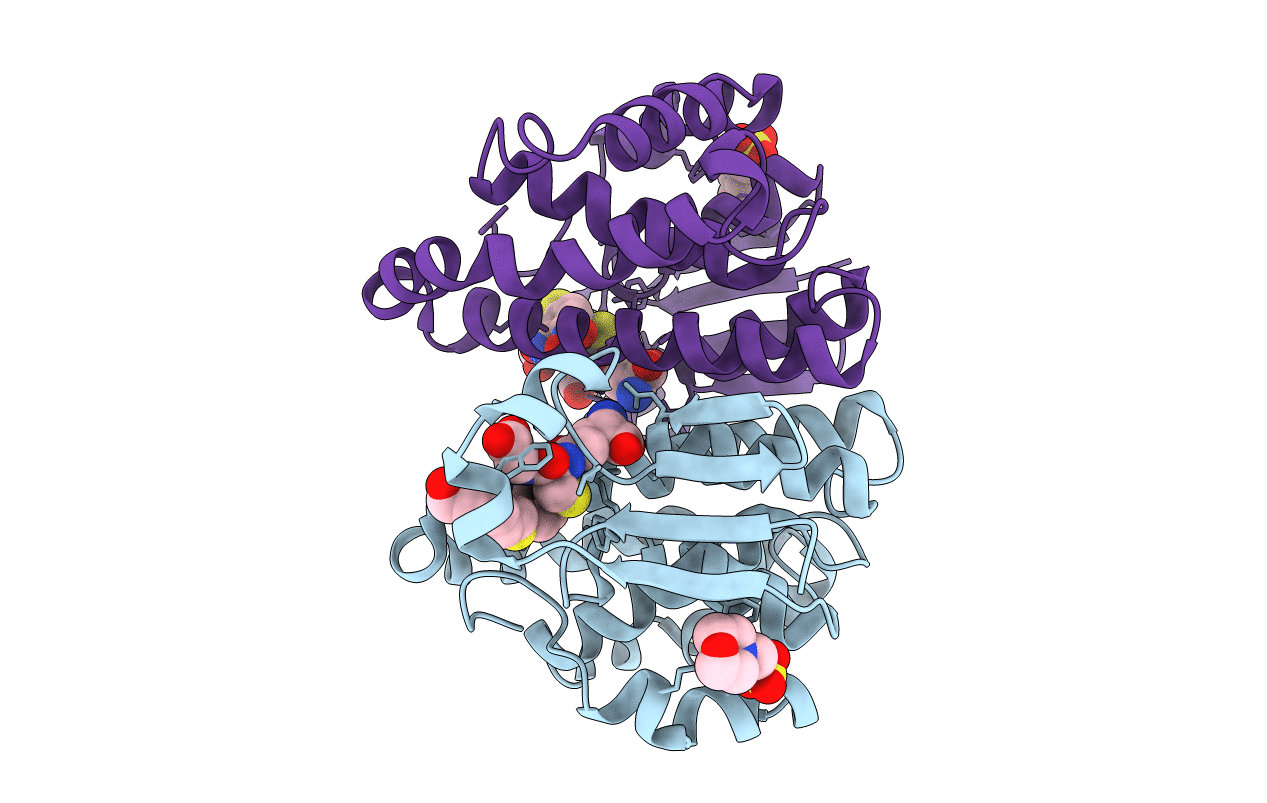
Deposition Date
2009-07-22
Release Date
2009-10-27
Last Version Date
2023-11-01
Entry Detail
PDB ID:
3IE3
Keywords:
Title:
Structural basis for the binding of the anti-cancer compound 6-(7-Nitro-2,1,3-benzoxadiazol-4-ylthio)hexanol (NBDHEX) to human glutathione S-transferases
Biological Source:
Source Organism(s):
Homo sapiens (Taxon ID: 9606)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
1.80 Å
R-Value Free:
0.20
R-Value Work:
0.17
R-Value Observed:
0.18
Space Group:
C 1 2 1


