Deposition Date
2008-09-30
Release Date
2009-03-24
Last Version Date
2023-09-06
Entry Detail
PDB ID:
3EPW
Keywords:
Title:
Crystal structure of Trypanosoma vivax nucleoside hydrolase in complex with the inhibitor (2R,3R,4S)-1-[(4-hydroxy-5H-pyrrolo[3,2-d]pyrimidin-7-yl)methyl]-2-(hydroxymethyl)pyrrolidin-3,4-diol
Biological Source:
Source Organism(s):
Trypanosoma vivax (Taxon ID: 5699)
Expression System(s):
Method Details:
Experimental Method:
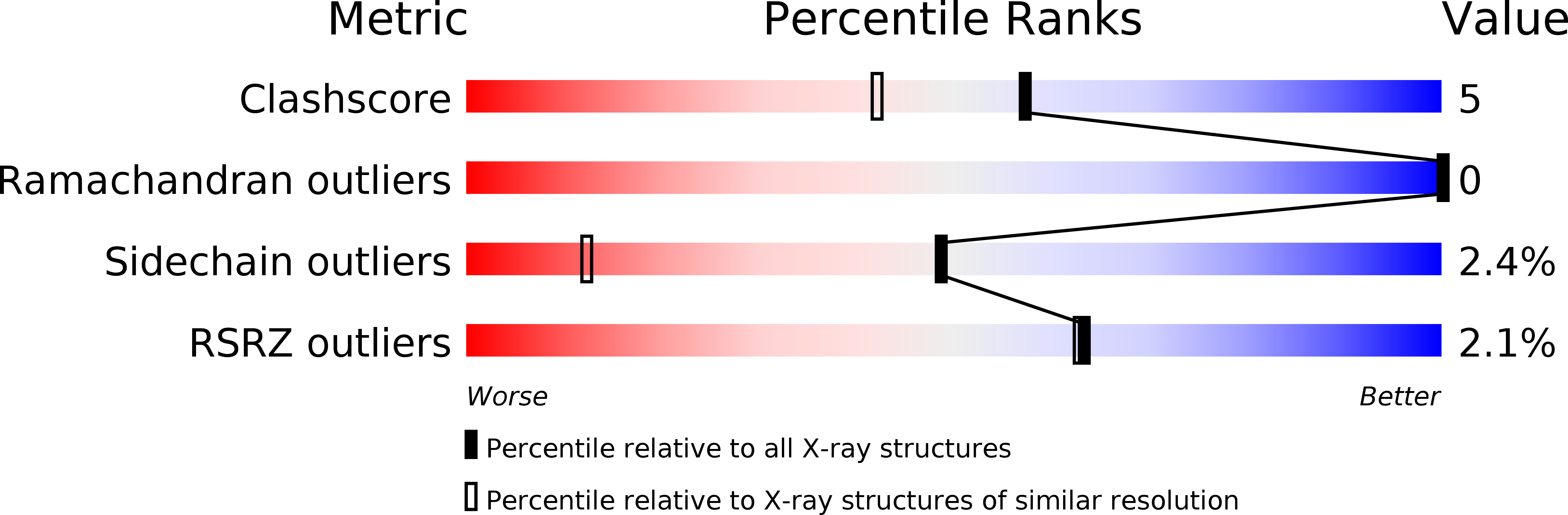
Resolution:
1.30 Å
R-Value Free:
0.18
R-Value Work:
0.16
R-Value Observed:
0.16
Space Group:
P 1 21 1