Deposition Date
2011-07-26
Release Date
2012-04-04
Last Version Date
2025-03-26
Entry Detail
PDB ID:
3B2C
Keywords:
Title:
Crystal structure of the collagen triple helix model [{PRO-HYP(R)-GLY}4-{HYP(S)-Pro-GLY}2-{PRO-HYP(R)-GLY}4]3
Method Details:
Experimental Method:
Resolution:
1.36 Å
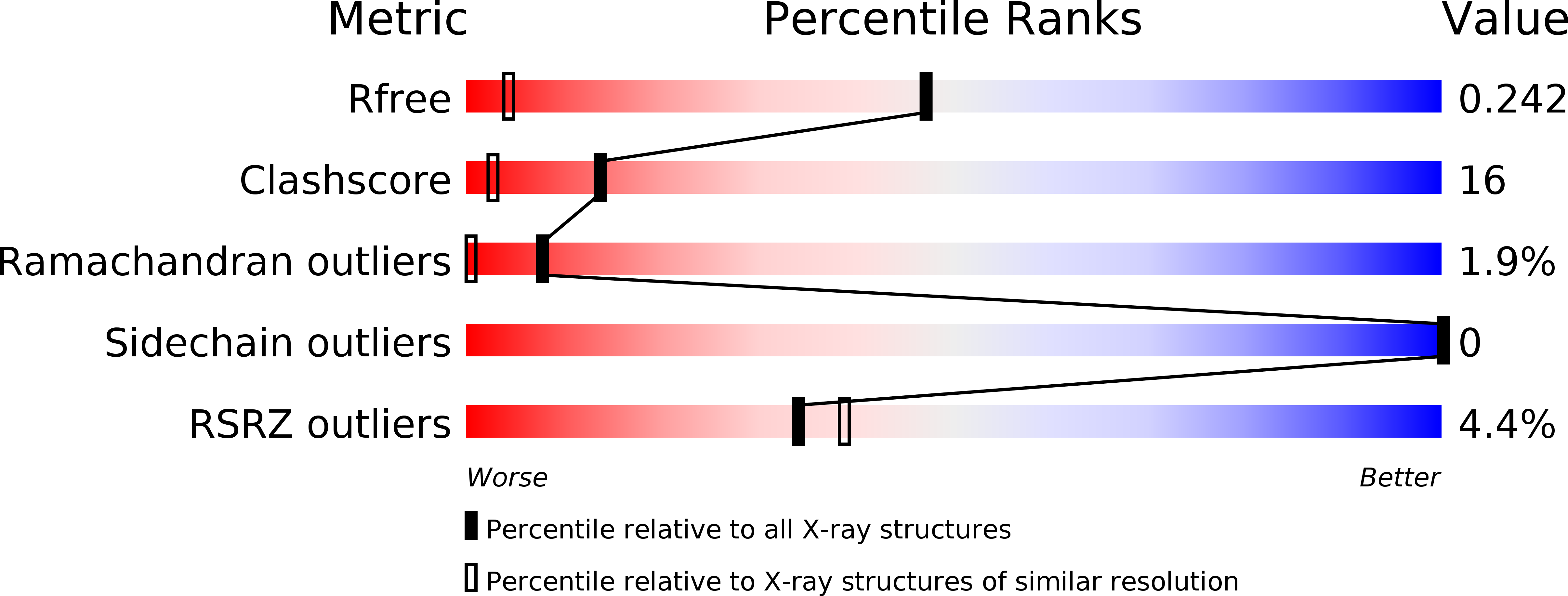
R-Value Free:
0.24
R-Value Work:
0.20
R-Value Observed:
0.20
Space Group:
P 1 21 1