Deposition Date
2010-06-05
Release Date
2011-04-20
Last Version Date
2024-10-16
Entry Detail
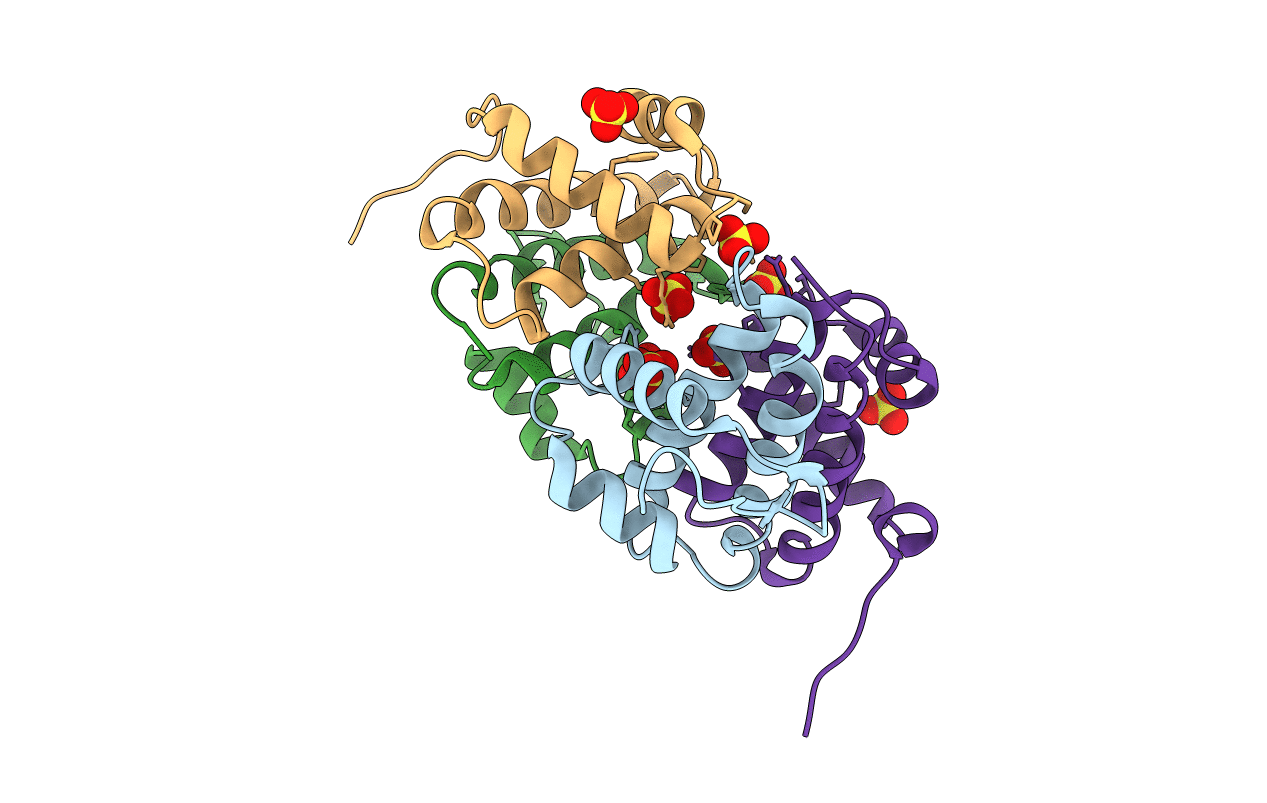
PDB ID:
3AJF
Keywords:
Title:
Structural insigths into dsRNA binding and RNA silencing suppression by NS3 protein of rice hoja blanca tenuivirus
Biological Source:
Source Organism(s):
Rice hoja blanca virus (Taxon ID: 480611)
Expression System(s):
Method Details:
Experimental Method:
Resolution:
2.00 Å
R-Value Free:
0.25
R-Value Work:
0.21
R-Value Observed:
0.21
Space Group:
P 41 21 2


