Deposition Date
2009-06-11
Release Date
2009-10-27
Last Version Date
2023-11-01
Entry Detail
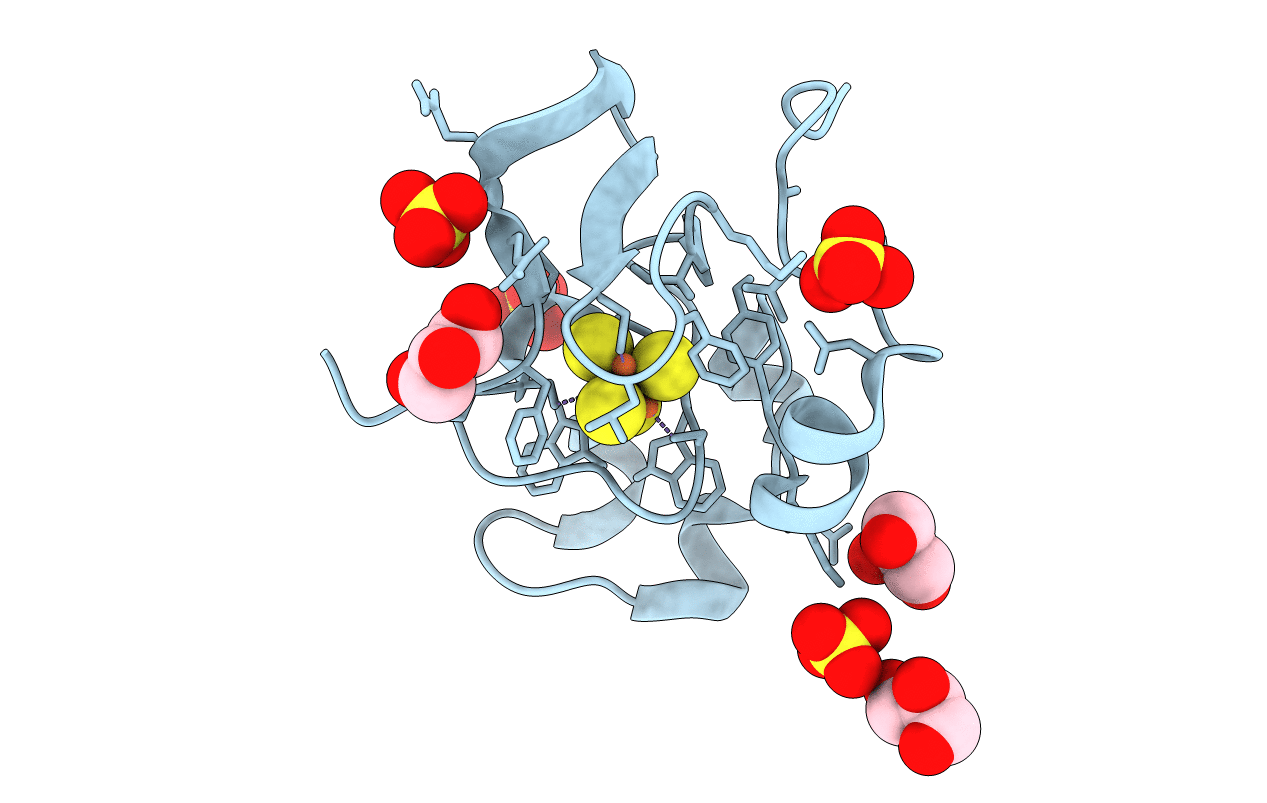
PDB ID:
3A39
Keywords:
Title:
Crystal Structure of High-Potential Iron-Sulfur Protein from Thermochromatium tepidum at 0.72 angstrom resolution
Biological Source:
Source Organism(s):
Thermochromatium tepidum (Taxon ID: 1050)
Method Details:
Experimental Method:
Resolution:
0.72 Å
R-Value Free:
0.07
R-Value Work:
0.06
R-Value Observed:
0.06
Space Group:
P 21 21 21


