Deposition Date
2007-06-25
Release Date
2008-04-08
Last Version Date
2024-10-16
Entry Detail
PDB ID:
2Z4T
Keywords:
Title:
Crystal Structure of Vibrionaceae Photobacterium sp. JT-ISH-224 2,6-sialyltransferase in a Ternary Complex with Donor Product CMP and Accepter Substrate Lactose
Biological Source:
Source Organism(s):
Photobacterium sp. (Taxon ID: 426532)
Expression System(s):
Method Details:
Experimental Method:
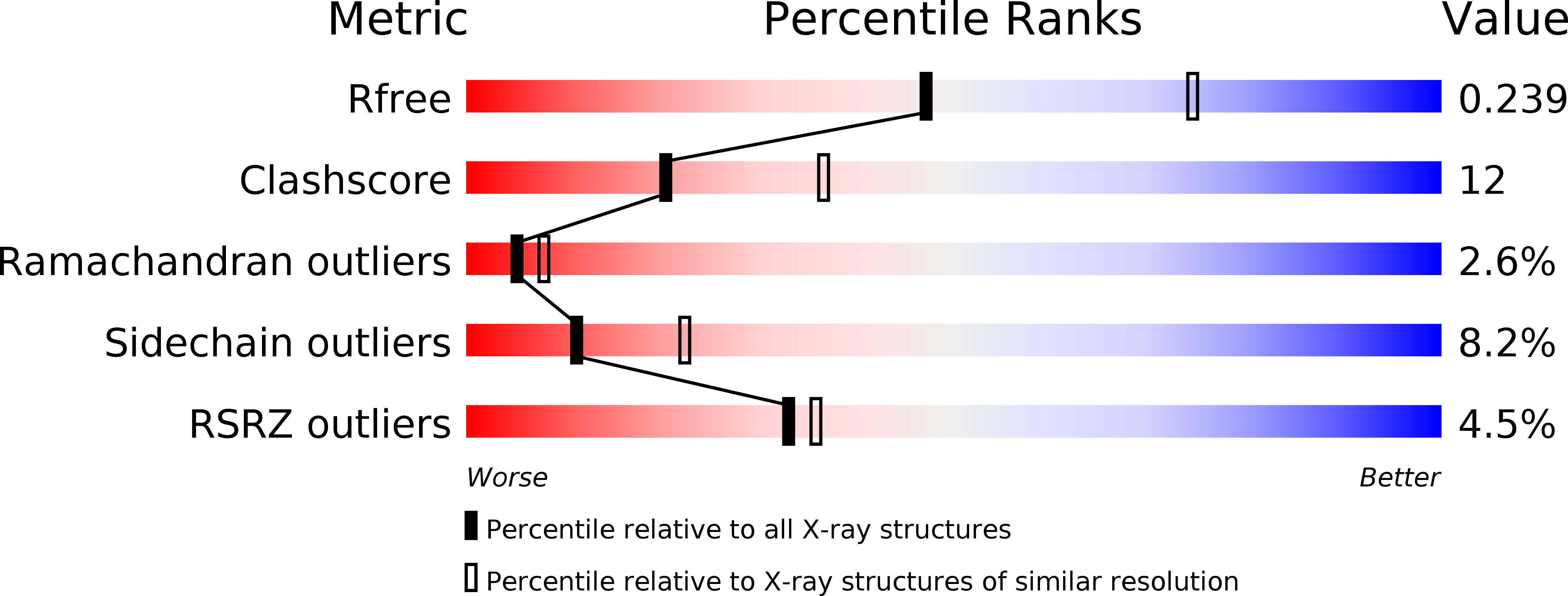
Resolution:
2.50 Å
R-Value Free:
0.24
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 31 2 1