Deposition Date
2010-05-10
Release Date
2010-05-19
Last Version Date
2023-12-20
Entry Detail
PDB ID:
2XE1
Keywords:
Title:
Molecular insights into clinically isolated OmpC mutants and their role in multi-drug resistance
Biological Source:
Source Organism(s):
ESCHERICHIA COLI (Taxon ID: 562)
Expression System(s):
Method Details:
Experimental Method:
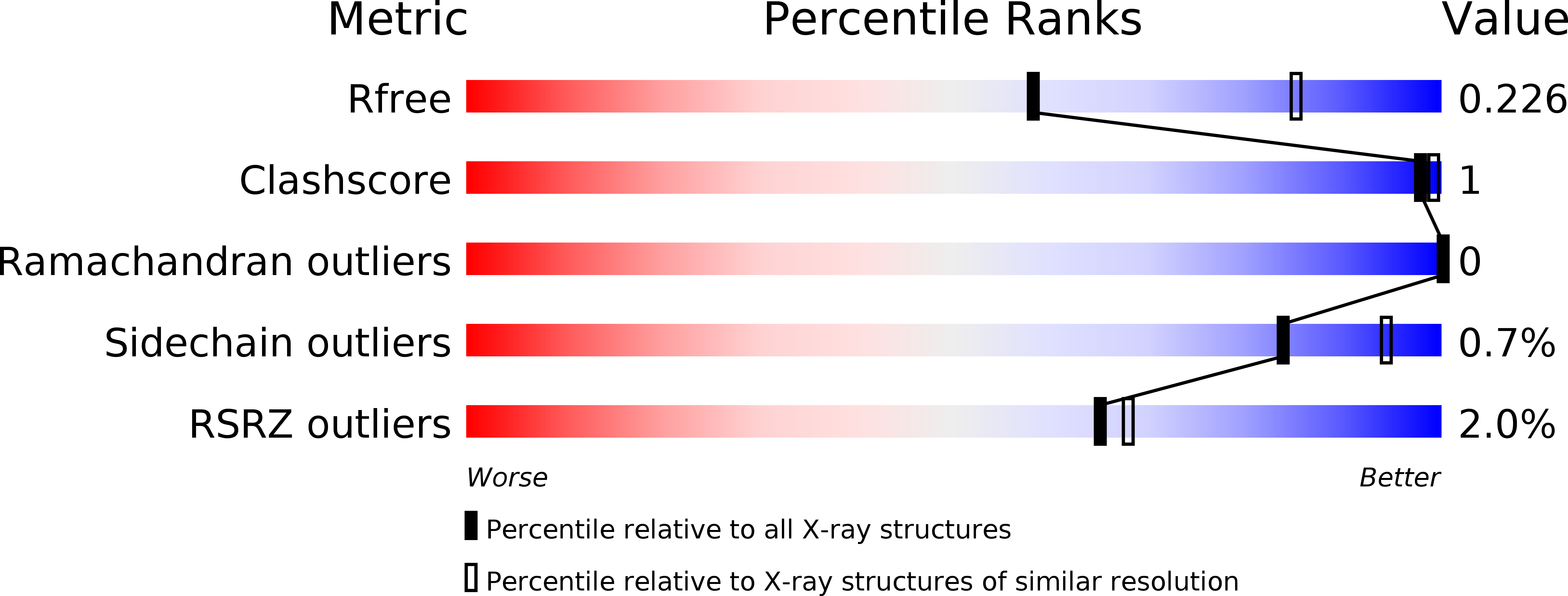
Resolution:
2.50 Å
R-Value Free:
0.21
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
P 63 2 2