Deposition Date
2010-04-15
Release Date
2011-04-06
Last Version Date
2023-12-20
Entry Detail
PDB ID:
2XBT
Keywords:
Title:
Structure of a scaffoldin carbohydrate-binding module family 3b from the cellulosome of Bacteroides cellulosolvens: Structural diversity and implications for carbohydrate binding
Biological Source:
Source Organism(s):
BACTEROIDES CELLULOSOLVENS (Taxon ID: 35825)
Expression System(s):
Method Details:
Experimental Method:
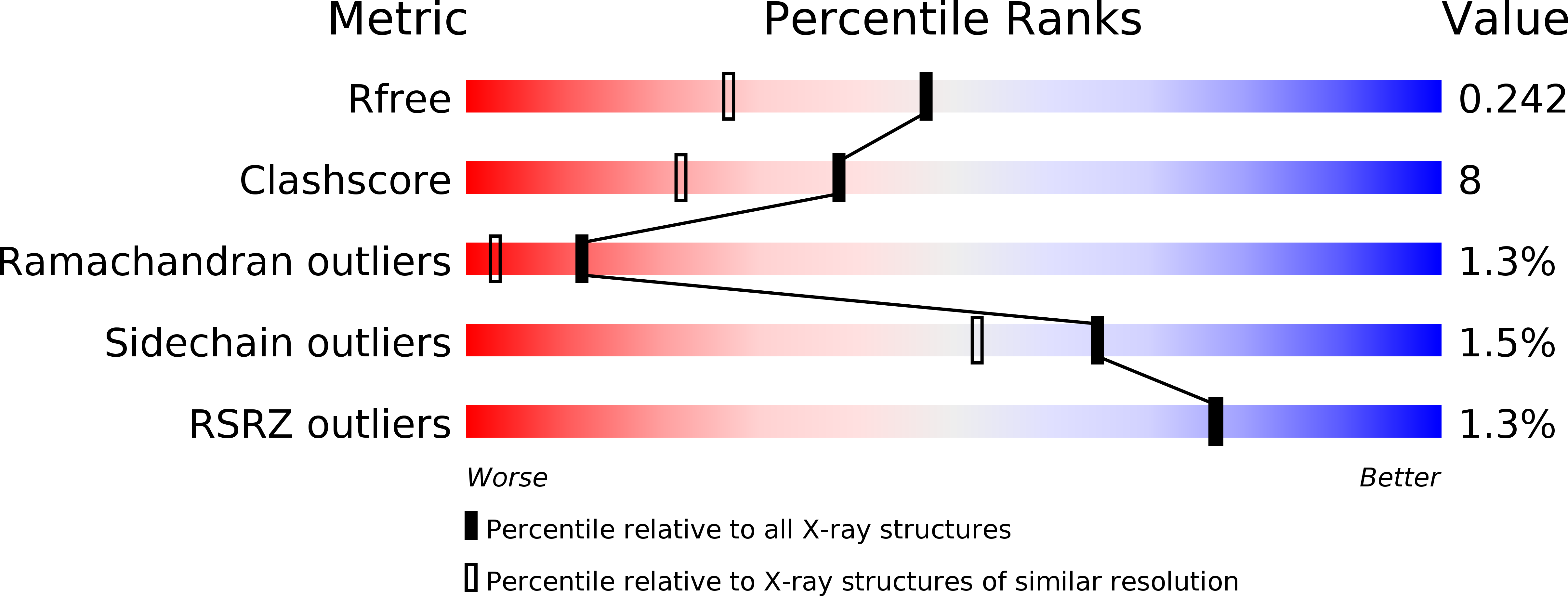
Resolution:
1.83 Å
R-Value Free:
0.24
R-Value Work:
0.19
R-Value Observed:
0.19
Space Group:
I 41 2 2