Deposition Date
2007-02-02
Release Date
2008-02-12
Last Version Date
2024-11-13
Entry Detail
PDB ID:
2OR9
Keywords:
Title:
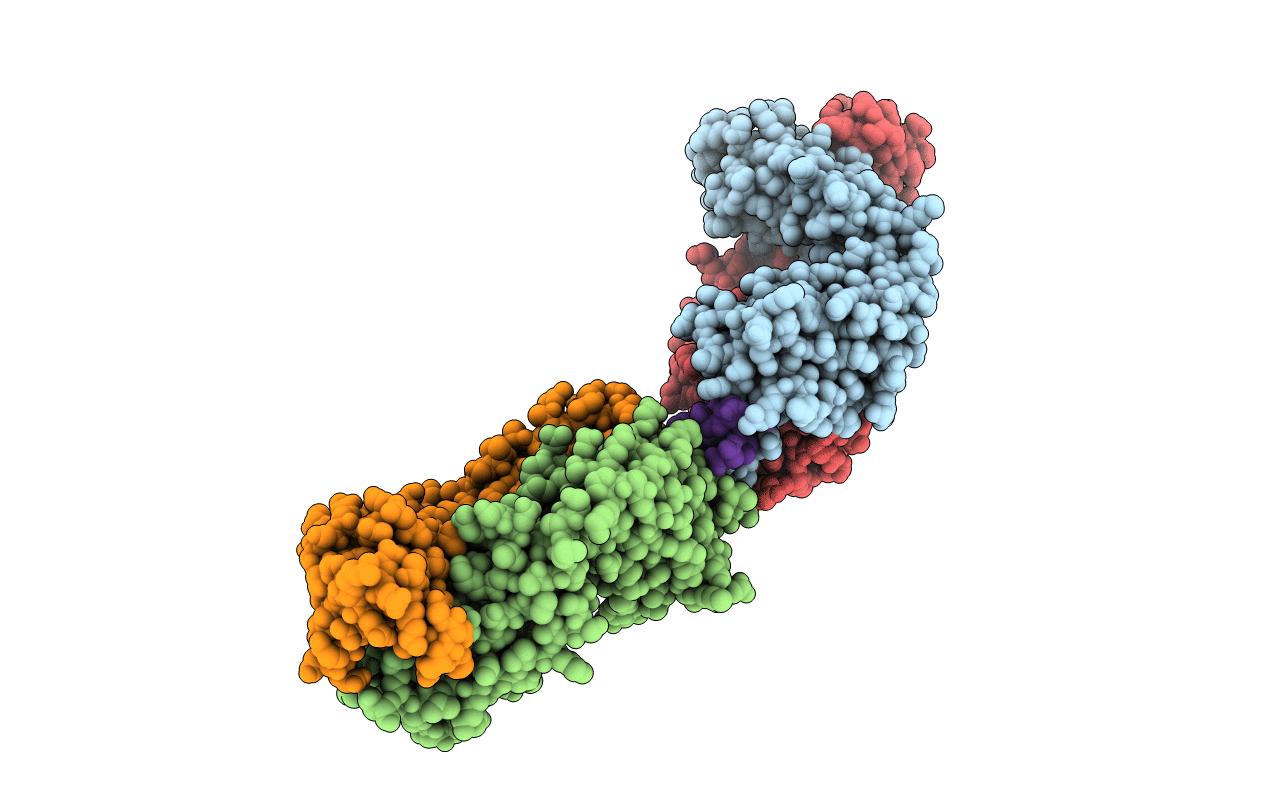
The structure of the anti-c-myc antibody 9E10 Fab fragment/epitope peptide complex reveals a novel binding mode dominated by the heavy chain hypervariable loops
Biological Source:
Source Organism(s):
Mus musculus (Taxon ID: 10090)
Method Details:
Experimental Method:
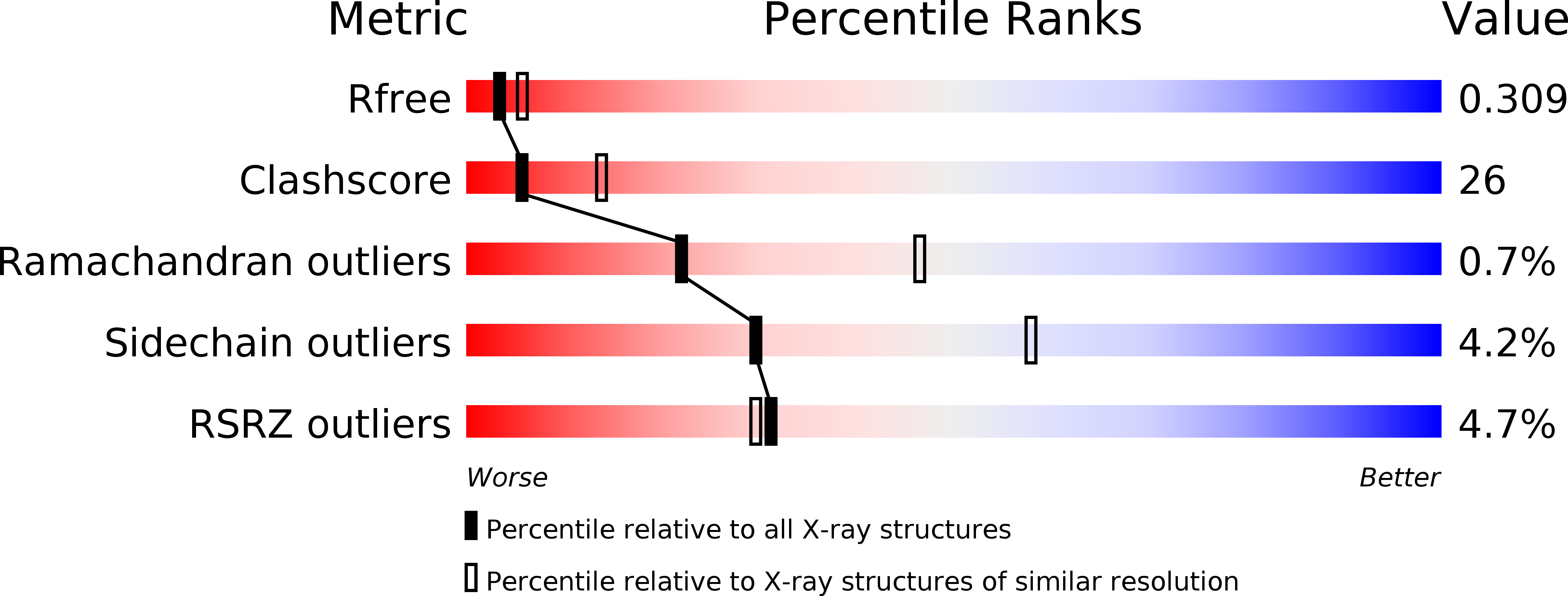
Resolution:
2.70 Å
R-Value Free:
0.31
R-Value Work:
0.25
Space Group:
P 21 21 21