Deposition Date
2013-10-11
Release Date
2014-02-26
Last Version Date
2024-05-01
Entry Detail
PDB ID:
2MFG
Keywords:
Title:
Csr/Rsm protein-RNA recognition - A molecular affinity ruler: RsmZ(SL4)/RsmE(dimer) 2:1 complex
Biological Source:
Source Organism(s):
Pseudomonas fluorescens (Taxon ID: 294)
Expression System(s):
Method Details:
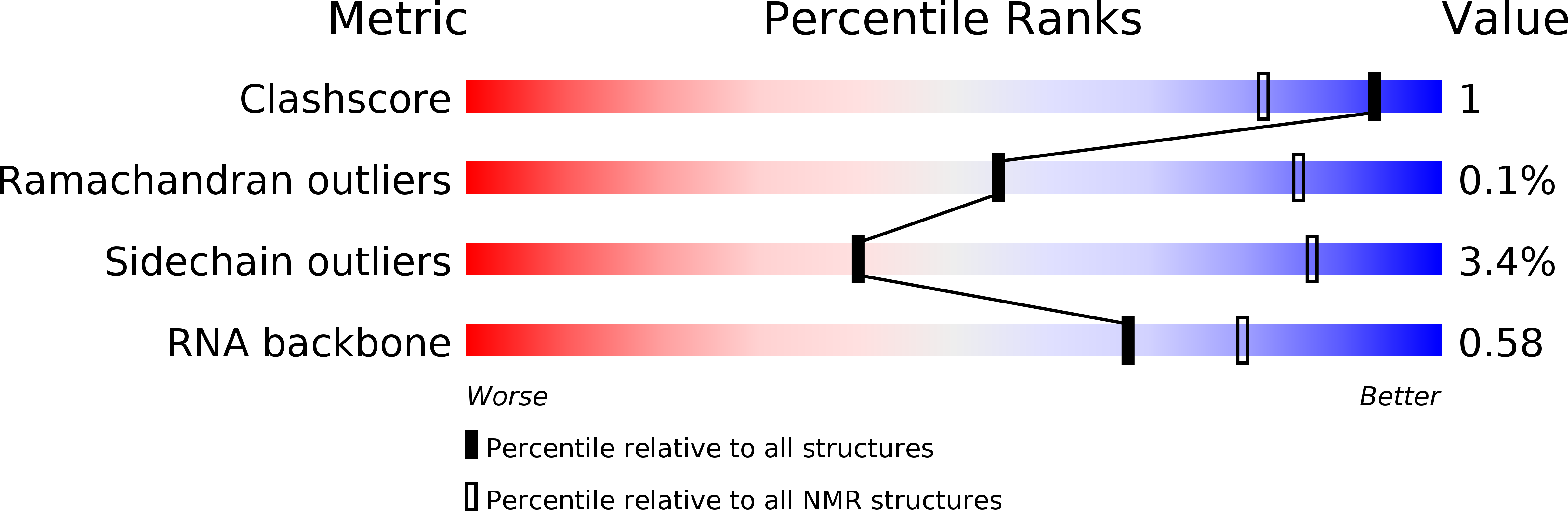
Experimental Method:
Conformers Calculated:
999
Conformers Submitted:
20
Selection Criteria:
structures with the lowest energy